-
磁性液体是粒径在10 nm左右的单畴磁性纳米颗粒分散于非磁性基液中构成的胶体悬浮体[1-2],为液态的磁功能材料.为了避免磁性纳米颗粒的自发团聚,可采用使颗粒表面带电荷或颗粒包裹表面活性剂的方法,通过静电排斥或空间位阻效应使颗粒保持分散性.前者叫离子型液体,后者叫表面型磁性液体.磁性液体中的磁性纳米颗粒的尺寸很小,其Brownian运动可有效地克服沉淀作用,并使磁性液体在没有外磁场作用下呈光学各向同性.在外磁场作用下,磁性液体中的磁性纳米颗粒的磁矩趋于磁场方向,从而使颗粒间的磁相互作用增强,形成链状团聚体,以致磁性液体呈光学各向异性.于是,当光束垂直磁场透过磁性液体样品时,将产生双折射效应和二向色性,称之为磁双折射和磁二向色性.磁双折射和磁二向色性是磁性液体主要被研究和应用的磁光效应[3-4].磁双折射Δn是振动方向垂直与平行磁场的偏振光的折射率差异,可表示为:
式中:λ是入射光波长,l是样品厚度,Δτ是垂直于平行磁场方向的偏振光透射磁性液体样品后的位相差.
磁二向色性是垂直与平行磁场方向的偏振光的衰减差Δκ.对于磁性液体,由于散射比吸收弱得多.因而衰减可表示为吸收差异,当强度为I0光束透过样品后,其透射强度为It,两者的差异为I=I0e-α,其中α为吸收度.因此,磁二向色性可表示为
式中I//和I⊥分别是偏振方向平行和垂直磁场方向的透射光的强度.通常采用线偏振光研究磁双折射和磁二向色性[5-6].然而这样的方法不能同时测得磁双折射和磁二向色性.
γ-Fe2O3纳米颗粒是合成磁性液体常用的一种磁性纳米颗粒.由液相合成γ-Fe2O3颗粒通常需要3个甚至更多的步骤[7].在前期工作中,我们提出了将FeOOH/Mg(OH)2前驱体经FeCl2溶液处理制备磁性纳米颗粒的化学诱导相变法[8-9].实验表明,用此方法可制备由FeCl3包裹的γ-Fe2O3纳米颗粒,这种纳米颗粒可用于合成离子型磁性液体[10].此外,我们提出了用圆偏振光研究磁性液体磁光效应的方法[11].在这种测试方法中,磁双折射和磁二向色性可同时确定.本研究尝试对化学诱导相变法制备γ-Fe2O3纳米颗粒进行油酸包裹以合成表面型磁性液体,发展了用圆偏振光方法测量磁双折射和磁二向色性的θ扫描技术.
HTML
-
将Mg(NO3)2溶液(2 mol/mL,10 mL)与FeCl3溶液(1 mol/mL,40 mL)混合,再与NaOH溶液(0.7 mol/mL,500 mL)混合.混合液在搅拌条件下加热至沸腾,并保持沸腾5 min.停止加热后自然冷却至室温.将沉淀物清洗至上部清液的pH值为7~8,然后倒掉上部清液.
-
将前驱体加蒸馏水至250 mL,充分混合后然后再与FeCl2溶液(0.087 mol/mL,550 mL)混合加热至70 ℃,保温30 min.停止加热后通过磁分离得到γ-Fe2O3纳米颗粒沉淀物.将此沉淀物与400 mL蒸馏水混合加热至70 ℃,再加入4 mL油酸,70 ℃保温1 h.用蒸馏水清洗,再经用磁分离得到油酸包裹的γ-Fe2O3沉淀物.最后经真空抽滤、干燥、研磨得到γ-Fe2O3磁性纳米颗粒.
-
称取油酸包裹的磁性纳米颗粒的质量为m(单位:g).根据所需颗粒体积分数ϕv,可计算所需煤油的加入量V(单位:mL)为
式中:ρc=2.72 g/cm3为油酸包裹的磁性纳米颗粒的平均密度[12].将颗粒与煤油混合后加热至110 ℃,保温1 h.停止加热后自然冷却至室温.分别配制了ϕv=0.4%,1.2%和2.0% 3种颗粒体积分数的磁性液体样品.
-
用HH-15振动样品磁强计(VSM)测量了油酸包裹的γ-Fe2O3磁性纳米颗粒的比磁化曲线.用TecaiG20ST透射电子显微镜(TEM)观察了颗粒的形态,并测定了其粒径.
1.1. 前驱体的制备
1.2. 磁性纳米颗粒的制备
1.3. 煤油基磁性液体的合成
1.4. 颗粒的表征
-
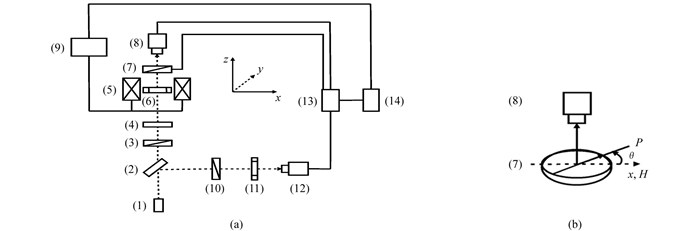
使用LabVIEW语言开发了通过圆偏振光测量磁性液体的磁双折射和磁二向色性的θ扫描技术.测试系统的示意图见图 1.在该系统中,可通过计算机记录和分别来自测量光路的光电探头(8)和参考光路中的光电探头(12)的信号.在加场测量前,首先通过偏振器(10)调整光电探头(8)和(12)测得的光强度相同为Ib.加场测量时,可得到T(θ)=It(θ)/Ib(It(θ)为由分析器(7)在θ角测得的强度),即
式中:I0为入射光的光强度.因此T(θ)是与θ有关的加场后、前的相对透射率.于是,通过检偏器(7)的转动进行θ扫描可测得T-θ曲线.在该曲线中,θ=0为磁场方向;θ=90°为与场垂直的方向.
本实验中使用了He-Ne激光器,其波长为λ=632.8 nm,玻璃样品盒的厚度为0.3 mm.
-
一束圆偏振光透过各向异性介质后,其透射光将变成椭圆偏振光.从椭圆的长度和取向可以获知介质的光学性质[13].在磁场作用下,磁性液体中的单畴磁性纳米颗粒将沿场方向形成链状结构,以致平行场方向和垂直场方向的折射率和吸收度不同,呈现磁双折射效应Δn和磁二向色性Δκ.导致双折射效应的位相差Δτ以及导致二向色性的吸收度差Δα可分别表示为[11]:
式中:
$ \tan \beta = \sqrt {\frac{{{T_{\min }}}}{{{T_{\max }}}}} $ ,Tmin和Tmax分别为T-θ曲线中T的最小值和最大值,对应透射椭圆的短、长轴;Δθ为T-θ曲线中Tmax对应的角度与90°之差,即对应透射椭圆的取向角.因此,当入射光源一定(即波长λ一定)和样品厚度l一定时,根据实验测得的T-θ曲线,可导出磁性液体样品的磁双折射效应Δn和磁二向色性Δκ.
2.1. 测量装置
2.2. 磁双折射和磁二向色性
-
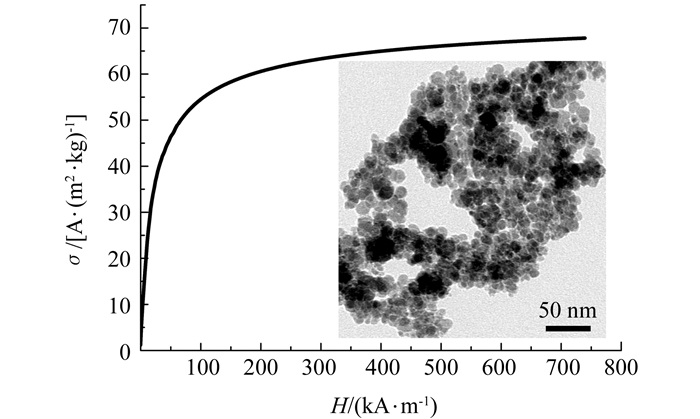
所制备的油酸包裹的γ-Fe2O3磁性纳米颗粒的比磁化曲线如图 2所示.由高场(H≥677 KA/m)的σ-1/H关系,导出其比饱和磁化强度[14]为:
TEM观察表明颗粒基本上为球形,如图 2中的插图所示.统计分析确定颗粒粒径呈对数正态分布,其中值粒径为xg=9.5 nm,标准偏差为lnσg=0.3.
-
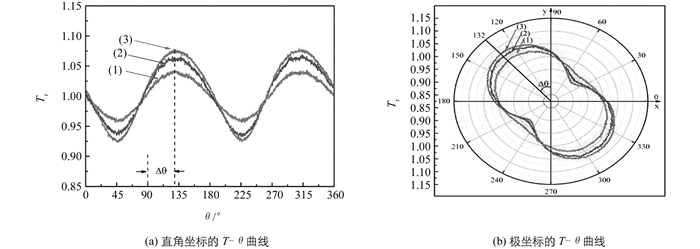
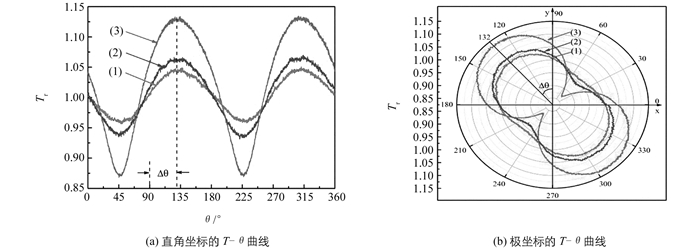
用右旋圆偏振光作为入射光,通过θ扫描技术得到的在不同条件下的透过磁性液体样品的相对透射率分布的T-θ曲线(图 3(a)和图 4(a)),曲线呈180°周期性.通过极坐标表示(图 3(b)和图 4(b)),可见T-θ曲线为闭合曲线,具有椭圆的180°对称性特点,表明在测量过程中,样品的场致微结构相变是稳定的.
-
从θ扫描的T-θ曲线得出的Tmin,Tmax和Δθ的实验数据见表 1和表 2.
根据实验得出的Tmin,Tmax和Δθ数据.由公式(5)和(6)可分别得出位相差Δτ和吸收度差Δα.进一步结合入射波长(λ=632.8 nm)及样品厚度(l=0.3 mm),由公式(1)和(2)最后得到反映磁双折射效应的Δn和反映磁二向色性的Δκ.这些磁光结果列于表 3和表 4中.
表 3和表 4的结果表明,所制备的γ-Fe2O3煤油基磁性液体,磁双折射效应Δn和磁二向色性Δκ随磁场强度的增强或颗粒体积分数的增加而增大.这个结果可解释如下:
γ-Fe2O3属介电各向同性的立方晶体,其球形纳米颗粒的光学性质是各向同性的,因此其分散的单颗粒体系不会产生磁双折射效应和磁二向色性[15].从TEM观察知,所制备的γ-Fe2O3煤油基磁性液体中的纳米颗粒是基本为球形的,因此其磁光效应来自于场致链结构而非单颗粒作用.在外磁场作用下,磁性液体样品中的γ-Fe2O3磁性纳米颗粒的磁矩趋于场方向,形成类链场致结构.当磁性液体的颗粒体积分数ϕv小于5%时,链间的磁相互作用可忽略,仅仅存在链中的近邻两颗粒的相互作用[16],随着外磁场强度的增强或随着磁性液体内部颗粒体积分数的增大,场致链的平均长度增长[17].因为磁双折射效应Δn和磁二向色性Δκ与链的平均长度成正比[18],所以当颗粒体积分数一定时(ϕv=1.2%),Δn和Δκ随磁场增强而增大;当磁场一定时(H=300/4π kA/m),Δn和Δκ随ϕv增强而增大.
3.1. 颗粒的表征
3.2. θ扫描的实验结果
3.3. 磁双折射效应和磁二向色性
-
1) 采用化学诱导相变制备的球形的γ-Fe2O3磁性纳米颗粒可通过油酸包裹合成煤油基磁性液体.
2) 由圆偏振光作为光源,通过θ扫描技术,可得到具有磁双折射效应Δn和磁二向色性Δκ信息的T-θ曲线.
3) 从T-θ曲线可判断样品的稳定性,从相关参数可解析得到的Δn和Δκ.
4) 所制备的γ-Fe2O3磁性液体的磁双折射效应和磁二向色性源自于场致链结构. Δn和Δκ随颗粒体积分数的增大或磁场强度的增强而增加.





 DownLoad:
DownLoad: