-
开放科学(资源服务)标识码(OSID):

-
慢性湿疹是一种慢性过敏性皮肤病,通常表现为持续性瘙痒、红疹、渗透、增厚、粗糙等[1]。慢性湿疹的发病机理复杂,主要由代谢异常、内分泌异常、外部环境中的病毒和细菌、温度与湿度的刺激和异物过敏原等[2-5]引起,因而造成一定程度的治疗困难。在外界因素刺激身体后,由于过敏原的出现,机体炎症失衡,细胞因子分泌紊乱和血管通透性改变,从而引发湿疹[6-7]。慢性湿疹的病因及发病机制还与免疫细胞和皮肤细胞的特征有关[8-9]。T细胞是慢性湿疹发生发展的关键有效因子[10-11]。通常,Th1和Th2细胞亚群保持动态平衡,但在某些因素的刺激下,Th1和Th2细胞中某一亚群功能升高,另一亚群功能降低,从而引起湿疹等炎症发生[12]。白三烯B4(leukotrienes,LTB4)是炎症中至关重要的一类炎性介质。LTB4可引起所有白细胞及其亚群向炎性组织浸润,加重Th2介导的炎症反应[13]。
黄连蜂房组主要由黄连与蜂房配伍而成,方中黄连具有清热解毒、燥湿润肤功效,湿敷时有收敛、止痒作用,能够迅速干燥创面、消炎、消肿、促进肉芽生长和创面修复[14]。研究表明黄连素能减少动物模型中炎症细胞的浸润[15]。此外,蜂房具有明显的抗肿瘤、抑菌、抗炎、治疗皮肤顽癣等作用[16]。蜂房还可用于治疗癣、痒、疹和银屑病[17]。尽管黄连和蜂房具有上述所说的一系列功效,但黄连与蜂房配伍能否发挥治疗慢性湿疹的功效及其作用机制尚未见报道。本研究拟初步探究黄连与蜂房配伍在治疗慢性湿疹中的抗炎作用及免疫机制,为治疗慢性湿疹提供新的治疗药物。
HTML
-
SPF级ICR小鼠(雄性,6~8周龄,20~25 g),购自北京华阜康生物科技股份有限公司。实验中所有的动物饲养均得到吉林农业大学动物伦理委员会许可(No:20231023,001)。
-
黄连与蜂房配伍提取液(自制);2,4-二硝基氯苯(DNCB)和丙酮(上海源叶生物技术有限公司);橄榄油(欧贝丽薇食用油品牌店);4%多聚甲醛PBS固定液(索莱宝科技有限公司);75%酒精(利尔康消毒科技有限公司);100 μL移液枪及枪头(上海微川精密仪器有限公司);组织匀浆机(河南郑州南北仪器有限公司,型号scientz-192);酶标仪(美国Thermo公司,型号MuLtiskan FC);台式高速冷冻离心机(法国Eppendorf公司,型号5804 R);涡旋振荡仪(其林贝尔仪器制造公司,型号QL-901);小鼠白细胞介素2,小鼠白细胞介素4,小鼠白三烯B4试剂盒(上海酶联生物科技有限公司);苏木精伊红(上海源叶生物技术有限公司)。LTB4RRabbit pAb(ABclonal,A15042);LTB4R2Rabbit pAb(ABclonal,A15479);β-Actin0028I102);polyclonalantibody(Bioworld,AP0060);Anti-RabbitIgG HRP Antibody(Millipore,AP132P);PVDF(millipore,IPVH00010);多功能成像系统(GE,LAS500);发光液(MILLIPORE,WBKLS0500);BCA蛋白浓度测定试剂盒(Thermo,23227);SYBR Master Mix(ThermoA25742);Primerscript RT reagent kit(ThermoM1631);Real-time PCR仪(ABI 7500);稳压电泳仪(Bio-Rad)。
-
50只SPF级ICR小鼠,随机分为空白对照组(CON)、模型组(M)、黄连组(C)、蜂房组(V)、黄连蜂房组(CV),每组10只。造模方法参照文献[18],各组小鼠实验前一天用剃须刀进行腹部剃毛,范围约为2 cm×2 cm。采用2,4-二硝基氯苯(DNCB)腹部致敏和耳部反复激发小鼠建立湿疹模型。除空白组外,其余组小鼠涂含有7% DNCB的丙酮橄榄油溶液(4∶1)100 μL致敏,末次致敏5 d后于小鼠双耳内外侧耳面均匀涂以0.5%DNCB溶液20 μL进行多次激发,1次/3 d,连续4次。空白组小鼠腹部及双耳均涂以丙酮橄榄油溶液(4∶1)对照,以相同的方式及相同的量进行涂抹。
-
各组于激发当天开始给药,若给药当天需进行DNCB激发,则给药时间与激发时间间隔2 h。空白对照组及模型组用蒸馏水涂抹腹部已脱毛皮肤及双耳内外侧;黄连组、蜂房组、黄连蜂房组分别在相同位置均匀涂抹100 mg的黄连提取液、蜂房提取液、黄连与蜂房配伍提取液,每日2次,连续12 d。
-
将取血后小鼠处死,摘取胸腺和脾,分别称质量并计算胸腺指数和脾指数。胸腺指数为每克体质量中胸腺的毫克数,脾指数为每克体质量中脾脏的毫克数。
-
小鼠耳朵皮肤组织标本经石蜡包埋固定,制成5 μm厚薄层切片,以苏木素-伊红(HE)进行染色,利用光学显微镜观察过敏反应过程中皮肤组织的病理变化,主要包括角化程度、真表皮水肿程度,以及炎症浸润等情况。
-
通过ELISA试剂盒对血清中的IL-2、IL-4及LTB4质量浓度进行检测。重复3次实验。
-
采用Trizol法抽提细胞中的总RNA,根据试剂盒进行反转录成cDNA。反应条件和引物序列见表 1和表 2。通过RT-PCR检测细胞样品中目的基因和内参基因的表达量,根据RT-PCR反应曲线得到各样品目的基因和内参基因的Ct值(threshold cycle number阈值循环数),采用ΔΔCt的方法进行相对定量。
-
小鼠皮肤组织匀浆液用全细胞裂解液裂解细胞,使细胞全部破裂,充分裂解后无明显沉淀,4 ℃ 12 000r/min离心10 min,取上清,用BCA法测定蛋白浓度。然后进行制胶、点样、电泳、转膜、封闭、洗膜,取出膜在TBST中洗涤3次,10 min/次;将膜放入抗体孵育盒,加入适当浓度的一抗,4 ℃过夜。取出膜在TBST中洗涤3次,10 min/次;将膜放入抗体孵育盒,根据说明书加入适当浓度的二抗,室温孵育1 h。取出膜在TBST中洗涤3次,10 min/次;将膜放入暗盒中,滴加ECL反应液(A和B液等体积混合后立即使用),用化学发光成像系统扫描,并根据显影情况调整系统曝光时间,导出数据。重复3次实验。
-
用Graphpad Prism 10.1.2软件对所有数据进行分析,计算出相应的统计学参数。
1.1. 供试材料
1.2. 试剂与仪器
1.3. 体外实验方法
1.3.1. 分组与造模
1.3.2. 给药
1.3.3. 免疫器官指数检测
1.3.4. HE染色法观察皮肤组织的病理变化
1.3.5. ELISA法检测血清中炎症因子的质量浓度
1.3.6. RT-PCR法检测皮肤组织BLT1和BLT2 mRNA的表达
1.3.7. Western blot法检测皮肤组织BLT1和BLT2蛋白的表达
1.4. 统计方法
-
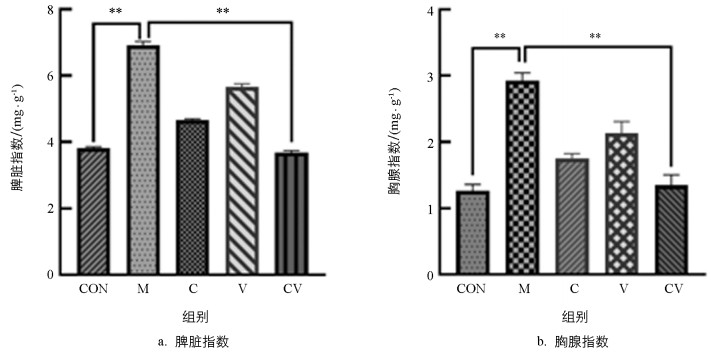
黄连与蜂房配伍对慢性湿疹小鼠脾脏和胸腺的影响如图 1所示:与空白对照组相比,模型组小鼠脾指数和胸腺指数极显著升高,且差异都极显著(p<0.01);与模型组比较,黄连蜂房组小鼠的脾指数和胸腺指数极显著降低(p<0.01)。
-
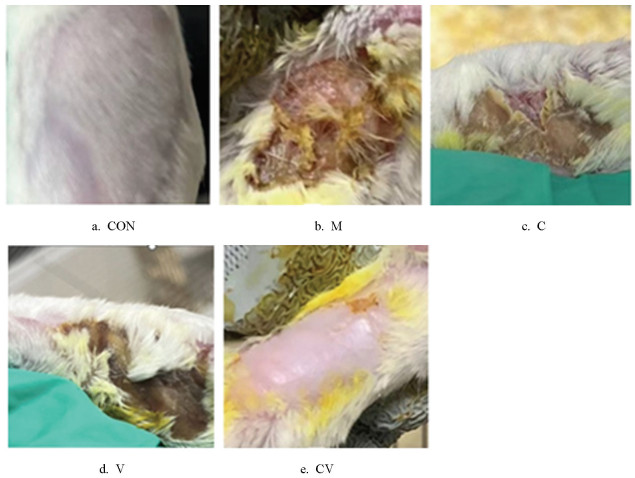
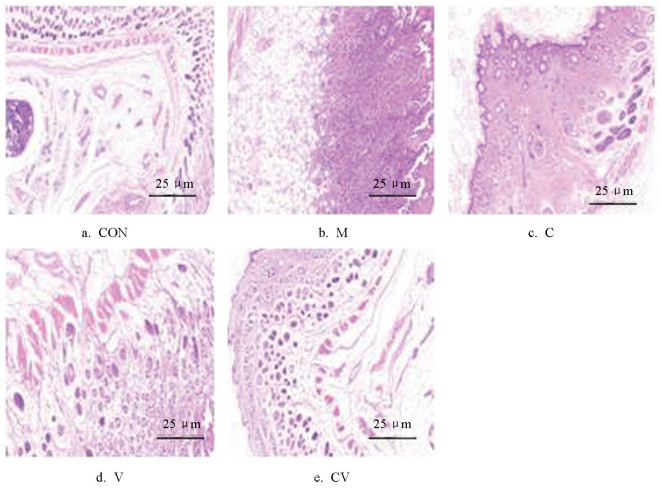
黄连与蜂房配伍对慢性湿疹小鼠皮肤病理表现的影响如图 2、3所示。空白对照组小鼠的皮肤保持完整,组织结构各层次分明,表皮、真皮和皮下组织之间的界限清晰,且角质层厚度适中,透明且光滑。与空白对照组相比,模型组小鼠皮损明显,表皮细胞呈现出显著的增生现象,细胞排列紊乱,失去了正常的层次结构,同时,角化过度的问题也十分突出。此外,内皮细胞出现了水肿和充血,提示有局部血流被动充盈,这表明小鼠的皮肤可能受到过度的炎症反应。黄连组和蜂房组小鼠的皮肤组织结构逐渐恢复,小鼠的皮损明显减轻;黄连蜂房组小鼠的治疗效果更为显著,小鼠皮损部位角化程度显著降低,炎性细胞的浸润量显著减少,损伤程度较模型组明显改善。
-
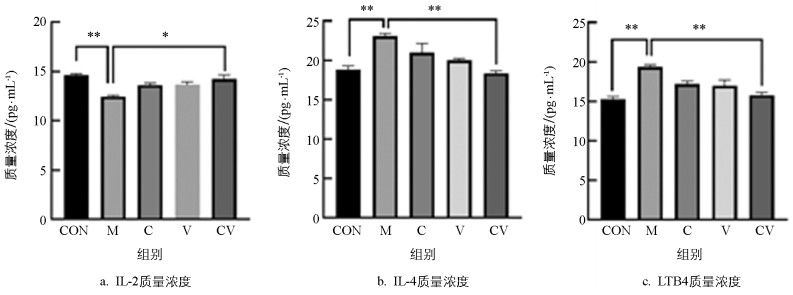
IL-4是Th2细胞分泌的细胞因子之一,IL-4能够促进B细胞生成IgE,从而加剧慢性湿疹炎症反应。LTB4能够刺激促炎症细胞介质与因子,参与慢性湿疹的发生。慢性湿疹模型小鼠组织和血清中的IL-4和LTB4水平显著高于空白对照组[19]。与空白组相比,慢性湿疹模型对照组的血清IL-2水平下降,而IL-4水平升高[20]。
如图 4所示:与空白对照组相比,模型组小鼠血清中IL-4、LTB4极显著升高,IL-2极显著降低,具有极显著性差异(p<0.01);与模型组相比,黄连蜂房组小鼠血清中IL-4、LTB4极显著降低(p<0.01),IL-2显著升高(p<0.05)。
-
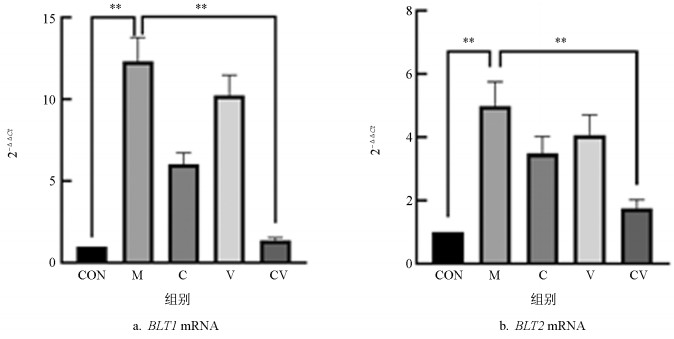
由图 5a可知:与空白对照组相比,模型组小鼠皮肤组织BLT1 mRNA的表达水平有极显著提高(p<0.01);与模型组相比,黄连蜂房组对慢性湿疹小鼠皮损组织BLT1 mRNA的表达有极显著的抑制作用(p<0.01)。
根据图 5b可知:与空白对照组相比,模型组小鼠皮肤组织BLT2 mRNA的表达水平有极显著提高(p<0.01);与模型组相比,黄连蜂房组对慢性湿疹小鼠皮损组织BLT2 mRNA的表达有极显著的抑制作用(p<0.01)。
-
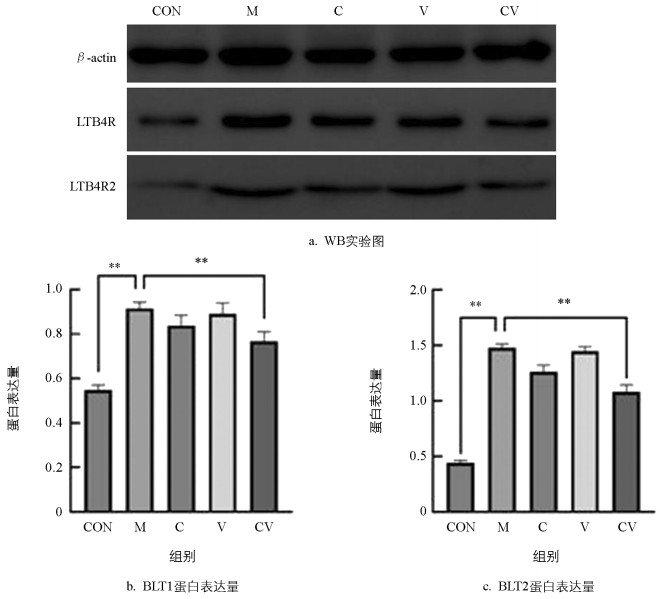
用ImageJ软件对WB条带进行灰度分析,以β-actin为内参蛋白,分别计算LTB4R/β-actin和LTB4R2/β-actin的比值,WB结果(图 6)显示:与空白对照组相比,LTB4R和LTB4R2蛋白在模型组慢性湿疹小鼠中的表达极显著增高(p<0.01);与模型组相比,LTB4R蛋白和LTB4R2蛋白在黄连蜂房组慢性湿疹小鼠中的表达极显著降低(p<0.01)。
2.1. 黄连与蜂房配伍对慢性湿疹小鼠免疫器官的影响
2.2. 黄连与蜂房配伍对慢性湿疹小鼠皮肤组织病理学变化的影响
2.3. 黄连与蜂房配伍对慢性湿疹小鼠血清中炎症因子分泌的影响
2.4. 黄连与蜂房配伍对慢性湿疹小鼠皮损组织BLT1和BLT2 mRNA表达的影响
2.5. 黄连与蜂房配伍对慢性湿疹小鼠皮损组织BLT1和BLT2蛋白表达的影响
-
我国一般人群的慢性湿疹患病率约为7.5%,儿童患病率可达18.71%[21]。由于其患病率高,病因复杂,病程长和治疗困难,严重影响了患者的生活质量[22-23]。慢性湿疹主要是由于Th1和Th2淋巴细胞介导的免疫失调,从而引起皮肤屏障功能障碍和瘙痒[24]。其中,Th1细胞主要分泌IL-2等炎症因子,主要介导细胞免疫反应,而且都是调节免疫反应的重要因素。而Th2细胞主要分泌IL-4、IL-5、IL-6等炎症因子,在介导体液免疫并在增强嗜中性白细胞介导的吞噬作用和细胞毒性中发挥良好作用[25]。因此选择IL-2和IL-4的含量作为检测指标来反映炎症因子的分泌平衡,探讨黄连与蜂房配伍对慢性湿疹小鼠的抗炎免疫机制。在本实验中,与空白对照组相比,模型组小鼠血清中IL-2显著降低,IL-4显著增加,表明Th1和Th2免疫失衡。黄连与蜂房配伍治疗组慢性湿疹小鼠血清中IL-4显著降低,IL-2显著升高,使Th1细胞与Th2细胞分泌的炎症因子达到平衡,从而达到治疗慢性湿疹的目的。
细胞免疫功能与慢性湿疹的发病有着重要的关系。胸腺是T细胞分化成熟的免疫中枢器官,脾脏是最大的外周免疫器官,成熟B淋巴细胞聚集于此,与细胞免疫关系密切。胸腺指数及脾指数是衡量机体免疫功能的初步指标[26]。慢性湿疹模型小鼠脾指数和胸腺指数均明显高于空白对照组,而黄连与蜂房配伍治疗组可极显著下调慢性湿疹小鼠的脾指数和胸腺指数,说明黄连与蜂房配伍可调节慢性湿疹模型小鼠的免疫功能从而达到治疗慢性湿疹的作用。
白三烯(LTs)是衍生自花生四烯酸的炎性介质。LTs包括二羟基酸LT(LTB4)和半胱氨酸LTs(CysLTs:LTC4,LTD4,LTE4),它们都参与急性和慢性炎症反应[27]。LTB4在许多炎症疾病、免疫系统疾病和变态反应方面发挥重要作用,LTB4的两个受体,分别为高亲和力的BLT1和低亲和力的BLT2,均属于G蛋白偶联受体[28]。药理学研究表明,BLT1信号刺激中性粒细胞的脱粒、趋化和吞噬作用,LTB4/BLT1信号通路的过度激活会引起急性和慢性炎症,包括哮喘[29]、特应性皮炎、牛皮癣、动脉粥样硬化[30]、关节炎、肥胖症[31]等。而BLT2在包括肠道和皮肤在内的各种组织的上皮细胞中高度表达。在皮肤损伤期间,表皮角质形成细胞上BLT2的激活通过增强细胞迁移来加速皮肤伤口愈合。在本实验中,黄连与蜂房配伍治疗组小鼠皮损组织中BLT1和BLT2 mRNA的表达得到极显著的抑制;LTB4R蛋白和LTB4R2蛋白在黄连蜂房组慢性湿疹小鼠中的表达极显著降低。
综上所述,黄连与蜂房配伍可以有效改善慢性湿疹皮肤病理状态,降低皮肤角化程度,减少炎性浸润,减少脾指数和胸腺指数,推测与黄连清热解毒、燥湿润肤、收敛、止痒,蜂房祛风、攻毒、杀虫、止痛的功效有关。





 DownLoad:
DownLoad: