-
开放科学(资源服务)标识码(OSID):

-
土壤是微生物重要的栖息地与优质的天然培养基,尤其在植物根际环境中,无论是微生物的群体特征分布与塑造,还是个体微生物与植物之间的相互关系,很大程度上受到了根系分泌物的调控[1]。植物通过根系将自身11%~40%的光合产物释放到根际环境中,营造出一个多样的生化环境,尤其能够影响根际微生物的聚集[1]。研究发现,根际微生物数量比原土体高5~50倍,在根中微生物的细胞总数达到1011个/g,且根际中更有利于富集细菌,真菌与细菌的生物量比值在根际土壤中为0.13~1.5,显著低于非根际土壤中的1.5~9.0[2]。根系所释放的分泌物如糖、有机酸、酶类等物质能够作为氮源、碳源或信号物质的定向选择或者刺激部分微生物在根部定殖,逐步构建适宜于自身稳定的根际微生物群落[3]。如黄瓜、番茄等作物释放的柠檬酸、琥珀酸、苹果酸能够显著选择性诱集部分微生物根部定殖与生长,且稳定同位素探针荧光标记方法证明了这些微生物能够利用这些根系分泌物[4]。
根系分泌物是一类重要的信息交流物质,推动着微生物与寄主植物互作协同进化,这种信号功能主要表现为趋化信号、共生信号、营养碳源,以及对有害生物的拮抗作用等[5]。尤其是趋化诱导信号,微生物利用自身趋化特性响应根系分泌物中的化学信号,选择生存环境,并快速定位寄主植物[6]。而关于根系分泌物趋化信号功能,Firmin等[2]早在1986年就首次证实了黄酮类物质在根瘤菌与豆科植物互作中的趋化信号作用。Turra等[8]发现尖孢镰刀菌(Fusarium oxysporum)通过趋性信号受体蛋白响应根系分泌物Class Ⅲ过氧化物酶进而定殖寄主植物根部,揭示了植物根系分泌物的信号功能以及真菌的趋性机制。目前,国内外较多学者已经聚焦于根际信号物质的挖掘与利用的研究,且大量根系分泌物逐步被证实为趋化信号物质,在微生物与寄主植物中起着重要作用。如羧酸类、芳香酸类等有机酸物质能够诱导恶臭假单胞菌(Pseudomonas putida)、铜绿假单胞杆菌(Pseudomonas aeruginosa)等的鞭毛运动与趋化活性[9]。Rudrappa等[10]研究发现,拟南芥叶部受到丁香假单胞菌(Pseudomonas syringae)侵染后,根系释放的L-苹果酸能够诱导枯草芽孢杆菌(Bacillus subtilis)的趋化活性与根部定殖。因此,了解根系分泌物的趋化信号功能,明确微生物在响应信号物质中的生物特性,对了解病害发生的机制及生物防治技术的研究具有重要意义与价值。基于此,本论文综述了根系分泌物的种类特征、趋化功能与机制,以及趋化信号的实践应用等,为进一步研究趋化信号提供重要的基础支撑。
HTML
-
根系分泌物是植物将部分光合产物通过根系释放到根际的一组复杂物质组合,包括低分子、高分子黏胶化合物,边缘细胞,离子以及植物组织的残落物等,种类达200余种[5]。而这些根系分泌物在植物与微生物互作过程中扮演着诸多重要的功能,且部分化合物的功能特征已经被证实。如柠檬酸、苹果酸、草酸等可通过鳌合、氧化还原等作用活化矿质养分[11],阿魏酸、香豆酸等酚酸物质能够作为化感物质,抑制其他植物的生长[12]。植物根尖能够释放出大量抗菌性化合物来抵御病原菌的侵染,如迷迭香酸、异黄酮类化合物、色氨酸衍生的次生代谢物、豌豆素等[13]。大麦在禾谷镰刀菌(Fusarium graminiarum)的侵染诱导下,根系能够分泌出阿魏酸、甲氧基肉桂酸、香草酸等大量苯丙烷类化合物,且这些化合物均能够显著抑制禾谷镰刀菌孢子萌发,对根部进行观察也发现在病原菌侵染后,根部外观颜色逐渐加深变黑,木质化程度逐渐加剧,表明植物在受到病原菌侵染后会合成并释放抗菌性化合物进行防御[14]。高分子有机物质如多糖、蛋白质以及一些磷脂质等,在根际环境中也具有根际健康保护的重要功能,如降低重金属的毒害作用,提高土壤团聚体的稳定性,防止土壤侵蚀,有助于根际的保水性[15]。根系代谢物除了抗菌防御与活化元素功能外,还具有多样的信号功能,趋避或引诱根际土壤微生物。如根系释放的低分子化合物碳水化合物、氨基酸以及植物激素能够吸引特定有益微生物进行根部防御[16-17]。尤其豆科植物根系释放的黄酮类化合物,能够作为信号物质,调控根瘤菌、丛枝真菌、外生真菌与寄主之间的共生系统,也是根瘤菌在豆科植物根部定殖、实现固氮的重要趋化信号物质[7]。此外,根系分泌物也能够激活根际微生物的群体变化,Yuan等[18]发现拟南芥在受到生物胁迫后,根系释放出长链有机酸物质,能够诱导有益微生物的根部聚集,提高自身防御机能。而禾本科植物根部释放苯并恶嗪类防御性次生代谢产物能够改变土壤微生物群,增强茉莉酮酸信号和植物防御[19]。
-
趋化性是微生物选择生存环境、快速定位寄主的一种重要的生物学特性。根系分泌物能够作为重要的趋化信号物质,诱导根际环境中微生物进行趋化反应,实现对寄主植物的有效定殖,最终形成对寄主植物的有效侵染和生物防御体系构建[6]。
-
植物相关微生物组是植物的第二基因组,在植物健康生长与抗逆防病体系的构建中起到至关重要的作用。在有益微生物与寄主植物互作中,根系分泌物是两者之间重要的桥梁纽带。多数有益微生物在寻找定殖寄主植物中,很大程度依赖根系分泌物的趋化信号作用[20]。目前已有较多根系分泌物种类被鉴定出能够诱导有益微生物的趋化活性,如拟南芥受到丁香假单胞菌DC3000侵染后,根系所释放的苹果酸能够诱导枯草芽孢杆菌趋化活性,并形成根部聚集,实现抗病防御[10]。芽孢杆菌属作为PGPRs家族的重要成员,广泛应用于促进植物发育与保护健康。研究发现,贝莱斯芽孢杆菌(Bacillus velezensis)SQR9能够响应黄瓜根系分泌物产生趋化反应,其中有机酸、氨基酸、糖类等39种化合物被鉴定为化学引诱剂[21]。Liu等[22]进一步研究发现,黄瓜根系分泌物中的D-半乳糖对SQR9具有较强的趋化引诱作用,并增强生物膜形成。胡小加等[23]的研究发现,巨大芽孢杆菌(Bacillus megatherium)A6对油菜根系分泌物中的6种有机酸、8种糖类具有较强趋化性,其中对苹果酸、山梨醇的趋化性最强。而枯草芽孢杆菌Tu-100对油菜根系分泌物中的氨基酸组分有趋化性,其中组氨酸、精氨酸、谷氨酸、天门冬氨酸等5种趋化性表现较强[24]。Gupta Sood等[25]发现根际有益菌荧光假单胞菌(Pseudomonas fluorescens)对番茄根系分泌物中的苹果酸与柠檬酸趋化性较强。苜蓿等豆科植物作为重要的饲料与油料作物,能够进行固氮共生,而其中主要机理在于豆科植物所释放的根系分泌物黄酮类、异丙黄酮类等能够显著地诱导根瘤菌趋化活性,定殖豆科植物根部,这是根瘤菌固氮的前提与第一步[20]。然而在根际环境中,由于根系分泌物组分复杂,诱导微生物趋化反应行为的因素多样,尤其根系分泌物浓度与距离根系范围的关系密切,这种浓度变化更进一步增加了趋化诱导的复杂性。
-
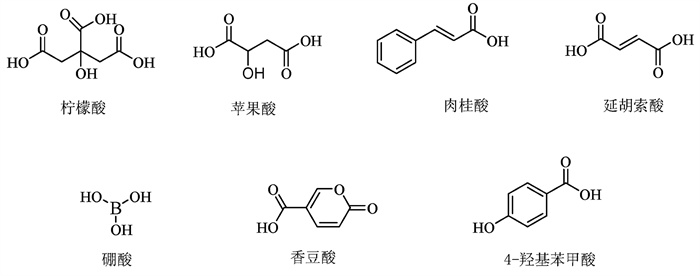
病原微生物在寄主植物侵染中,很大程度要依赖于自身趋化生物学特性,响应植物根际或叶际等部位释放出的化学信号物质,识别、定殖并进入寄主植物体内,最终形成侵染[6]。目前,已经有大量研究证实了根系分泌物中的信号物质能够显著诱导病原微生物趋化活性。例如,研究发现部分有机酸物质能作为土传病原细菌青枯菌(Rastonia solanacearum)的化学信号物质,诱导其趋化反应(图 1)。Yao等[26]发现番茄根系分泌物中的苹果酸、柠檬酸、琥珀酸对青枯菌K60具有显著趋化活性。Hasegawa等[27]系统地评估了番茄根系释放的30种芳香类有机酸,发现对香豆酸与4-羟基苯甲酸对青枯菌MAFF 730138有强烈的趋化诱导活性,并存在显著的浓度效应。在烟草根系分泌物中草酸、延胡索酸、肉桂酸能够显著地诱导青枯菌CQPS-1的趋化活性,并诱集其在烟株根部定殖[28]。此外,研究发现黄瓜、辣椒、甘蓝释放的根系分泌物草酸、乳酸能够诱导南方根结线虫(Meloidogyne incognita)的趋化反应,并诱集其在根部定殖[29]。
-
细菌的趋化系统是原核生物较为复杂的信号转导途径之一,由趋化受体蛋白(Methyl-accepting Chemotaxis Protein,MCP)、支架蛋白(Chew)、组氨酸激酶(CheA)、反应调节蛋白(CheY)、甲基酯酶(CheB)、甲基转移酶(CheR)、磷酸酶(CheC/CheX/CheZ),以及脱酰胺酶(CheD)等不同功能的蛋白组成[30]。趋化信号的转导机制主要是由MCPs负责接收并结合胞外趋化信号,MCPs结合信号物质后调控CheA的组氨酸激酶活性,将信号传递给下游的CheY,更进一步,CheA磷酸化的CheY通过鞭毛蛋白(FliM)与鞭毛-马达复合体相互作用,最终实现细菌运动方向的调控[30]。而在整个趋化反应过程中,MCPs与趋化物的特异性结合是细菌趋化反应形成的首要环节,也是微生物趋化反应中信号接收与转导的第一步,对于整个趋化过程至关重要。因此,明确趋化受体蛋白种类,是解析根系分泌物诱导微生物趋化活性机制的关键。近年来,很多学者成功鉴定出不同细菌中负责不同环境信号的MCPs,并对其生化特征进行了研究。如大肠杆菌(Escherichia coli)含有5种MCPs,分别是Tsr、Tar、Tap、Trg、Aer,能够与氨基酸、糖类稳定结合[30]。恶臭假单胞菌KT2440菌株基因组中含有27种MCPs蛋白[31],Lacal等[32]进一步研究发现该菌株的MCPs能够与苹果酸和延胡索酸稳定结合。Feng等[21]通过MCP基因敲除与回补的方法,证实了柠檬酸、苹果酸、草酸、富马酸等能够与解淀粉芽孢杆菌(Bacillus amyloliquefaciens)MCPA结合。Ni等[33]对丛毛单胞菌(Comamonas testosteroni)的趋化机理进行研究,通过构建20个MCP突变体,筛选并明确了MCP 2201能够与2,6-二羟基苯甲酸、2-羟基苯甲酸结合,且该蛋白被证实为第一个能够直接结合芳香化合物的甲基趋化受体蛋白。青枯菌GMI 1000已报道的MCPs蛋白有22种(含2种趋氧性蛋白),其中MCPA、MCPM与MCPB三个蛋白被证实能够与氨基酸、L-苹果酸、D-苹果酸、硼酸相结合,作为青枯菌PS 29的趋化信号物质[34-35]。
2.1. 根系分泌物对有益微生物的趋化诱导活性
2.2. 根系分泌物对病原微生物的趋化诱导活性
2.3. 根系分泌物诱导微生物趋化活性的机制
-
根系分泌物作为根际微生物与植物互作中重要的化学语言,能够通过诱导趋化反应,促进微生物快速定位识别寄主植物,实现有益微生物根部定殖、生防功能与根际健康调控。如根系分泌物中的苹果酸、草酸、柠檬酸等一些低分子化合物能够显著诱导根际促生细菌(PGPR)趋化活性,快速识别定殖寄主根部,实现根际健康调控与土传病害的有效控制[36-37]。在禾本科作物中,苯并噁唑嗪酮化合物是一类特有的根系分泌物,该类化合物兼具抗虫、除草等多种生物活性,尤其能够显著诱导有益菌恶臭假单胞菌的趋化活性,以及相关趋化运动基因的转录表达与根部定殖,还能够显著地吸引大量的有益微生物菌群,形成根部保护,抑制病原菌的侵染,这种化合物的存在可能是禾本科作物很少感染土传病害的原因[38, 19]。在拟南芥植物中,根系分泌物苹果酸能够诱导枯草芽孢杆菌的趋化活性与根部定殖,而将分泌苹果酸的基因突变后,发现枯草芽孢杆菌在根部定殖能力显著下降[10]。此外,根系分泌物在品种抗感病害差异中也扮演着重要的功能角色,抗病品种根系次生代谢物质能够吸引有益微生物菌群,形成良好的抗病生物屏障,提高对土传病害的控制。如不同抗感青枯病烟草品种根系分泌物富马酸、棕榈酸、苯甲酸等含量存在显著差异,而抗病品种K326根系分泌物能够显著诱导根际有益菌中型假食酸菌(Pseudacidovorax intermedius)和高卢根瘤菌(Rhizobium gallicum)的趋化活性,并实现对青枯菌的拮抗抑制与根际健康调控[39]。此外,利用作物根系分泌物种类特征的差异性,通过栽培方式可以有效降低病害发生。分蘖洋葱-番茄伴生模式体系下,分蘖洋葱根系释放的信号物质黄酮类化合物二氢槲皮素,能诱导番茄根部有益微生物Bacillus sp.的趋化活性与根际定殖,构建驱动抑病型根际微生物组,抑制番茄黄萎病[40]。
-
根系分泌物是土壤环境中重要的信号物质,在土壤-植物-微生物三者互作关系中扮演着营养、活化、抗性、趋化等信号功能角色,而趋化信号是微生物寻找、识别、定殖、共生以及侵染寄主植物的关键,探究趋化信号更有利于深入理解植物健康密钥、病害发生机制等,因此,该方面的研究已经成为国内外研究者关注的焦点之一。近年来,在根系分泌物趋化信号功能方面已经取得了部分研究进展,但由于根系分泌物种类繁多、数量微小、变化多样,且受植物品种、生育期、环境生物以及非生物因子等多种因素的影响[41],难以被精准检测出来,给挖掘更多有效的信号物质带来了巨大挑战。因此,借助于代谢组学、宏基因组学等技术探索趋化信号物质、了解趋化机制与过程,并利用信号研发新型绿色调控技术,对解决土传类病害、提升微生物定殖能力与生物防治水平具有重要的实践意义与价值。
根际趋化信号物质是微生物与寄主早期互作的重要媒介,通过激发植物释放出能够吸引有益微生物的根系分泌物,构建具有抑病或抗病功能的根际微生态环境,以实现植物健康调控[42]。在未来对于该领域研究可重点关注以下几个方面。①瞄准不同种类微生物,广泛挖掘具有趋化信号功能的代谢物种类,深入分析代谢物的代谢时期以及释放含量,并通过代谢组学分析其合成、转运与释放机制。②深入分析趋化信号物质与微生物之间的互作分子机制,更加深入地理解病害发生机制、有益菌的寄主定殖与生防机制。③开发与利用趋化信号物质,从病害发生角度,可通过调控或降解减少信号物质,切断信号途径来降低病原菌对寄主植物的识别与早期侵染。对于有益菌,可通过外源添加或者与生防菌协同使用,提高生防菌根部定殖效率,实现防治效果的提升与根际健康的保障。




 DownLoad:
DownLoad: