-
开放科学(资源服务)标志码(OSID):

-
锂离子电池(LIBs)被广泛应用于各种便携式电子设备中,虽性能已接近目前电极材料所能达到的极限,但仍不能满足人们对高容量的需求. 因而,开发高性能的锂离子电极材料成为当务之急. 自Nazar等人[1]第一次将金属磷化物用作锂离子电池负极材料后,研究者们制备了各种高容量、低极化的金属磷化物如FeP[2],Ni3P[3],Ni2P[4],Co2P[5]和CoP[6]等.
Ni2P属于富金属磷化物,其金属性较强,反应电位较低,是一种极具发展前景的锂离子负极材料和催化剂. 当Ni2P被用作锂离子电池负极材料时,其嵌锂和脱锂机理为:Ni2P+3Li++3e-↔Li3P+2Ni,在循环过程中,由于金属—磷(M—P)键断裂,其体积发生膨胀致使容量快速衰减[7]. 此外,Ni2P电导率较低,不利于Li+和电子的运输,从而导致其容量较低[8]. 可控合成具有特殊结构的纳米材料一方面可以缩短Li+扩散路径,增加电极材料与电解液的接触面积;另一方面,其特殊结构可缓解体积膨胀的问题,从而提高电极材料的循环性能. 例如,Ni2P纳米颗粒[9]、Ni2P纳米片[10]等纳米材料已经应用于锂离子电池,但是其性能仍有待改善. 同时,Ni2P与碳材料复合可以增加其电导率,加快Li+和电子的运输速率从而改善其储锂性能.
本研究以空心的介孔碳球(HMCSs)为载体,在其空腔内部及表面固载Ni(OH)2纳米片,然后通过低温磷化过程,合成具有核壳结构的Ni2P/C/Ni2P复合物. 其微观结构有以下优点:①具有大的比表面积,当其用作电极材料时与电解液的接触面积增大,有利于锂离子的运输;②其内部具有大的空腔,能够缓解其体积膨胀;③空心碳球能改善电极材料的导电性,并且具有弹性,可以缓解Ni2P颗粒在循环过程中的体积膨胀. 因此,本研究设计了一种新型Ni2P/C/Ni2P核壳结构,将其用作锂离子电池负极材料时,在电流密度100 mA/g下,循环200圈后,其容量保持在465 mAh/g. 故本研究中Ni2P/C/Ni2P核壳结构的制备为其他金属磷化物的制备及锂离子电池性能的提高提供了一种新方法.
HTML
-
空心的介孔碳球(HMCSs)的制备:正硅酸乙酯和间苯二酚甲醛低聚物(RF)共缩聚在二氧化硅(SiO2)纳米球上,以形成SiO2@RF. 其具体制备过程为:将3.46 mL正硅酸乙酯滴加到含70 mL乙醇、10 mL蒸馏水和3 mL氨水的混合溶液中,搅拌15 min. 接下来,在混合溶液中加入0.4 g间苯二酚和0.56 mL甲醛. 继续搅拌24 h后,将混合物离心,洗涤,并置于真空干燥箱中在70 ℃下烘干. 然后,将SiO2@RF在Ar气氛下,700 ℃,焙烧5 h,得到SiO2@C. 最后,HMCSs被获得,并在2 mol/L的氢氧化钠溶液中去除SiO2.
Ni(OH)2/C/Ni(OH)2的制备:首先,将50 mg硝酸镍、0.5 g尿素和0.5 g聚乙烯吡咯烷酮K-30溶于35 mL乙醇和5 mL蒸馏水的混合溶液中. 然后,将5 mg HMCSs超声分散到该混合溶液中. 超声1 h后,将该悬浊液转移到50 mL反应釜中,在120 ℃下反应12 h,待冷却到室温,离心,洗涤,干燥,得到Ni(OH)2/C/Ni(OH)2.
Ni2P/C/Ni2P的制备:将35 mg Ni(OH)2/C/Ni(OH)2与1.05 g NaH2PO2混合研磨成粉末. 然后,将该粉末在Ar气流下300 ℃退火3 h. 接下来,将退火后的产品用蒸馏水洗涤(直到彻底除去剩余的盐). 最后,在真空干燥箱中70 ℃干燥12 h得到Ni2P/C/Ni2P.
-
复合纳米材料的形貌及成分采用扫描电子显微镜(Hitachi,S-4800,Japan)、透射电子显微镜(Tecnai G2 F20 S-TWIN,USA)、X射线衍射仪(MAXima X XRD-7000,Japan)和显微共聚焦拉曼光谱仪(Renishaw Invia Reflex,UK)等仪器测试.
-
电极材料的电化学性能通过在手套箱中组装成CR2032型电池来研究. 扣式电池主要由参比电极(金属锂)、工作电极、隔膜(Celgard 2300)和电解液构成. 其工作电极的制备方法为:将质量比为7∶2∶1的电极材料、炭黑和羧甲基纤维素黏合剂(CMC)混合,并使其分散在蒸馏水中得到浆料,涂覆在铜箔上. 电解液是1 mol/L LiPF6溶于碳酸乙烯酯(EC)和碳酸二乙酯(DEC)中的混合溶剂(两者体积之比为1∶1).
恒电流充放电测试在蓝电测试仪上(Land CT2001A,China)进行,其测试条件:电压范围是0.005~3.0 V,电流密度为100 mA/g和1 000 mA/g. 循环伏安测试和电化学阻抗测试在电化学工作站(CHI 660d电化学工作站)进行测试,其测试条件:电压范围是0.005~3.0 V,扫描速率为0.5 mV/s. 阻抗测试条件:频率范围为0.1~10 000 Hz.
1.1. Ni2P/C/Ni2P的制备
1.2. 材料的表征
1.3. 电池的组装与测试条件
-
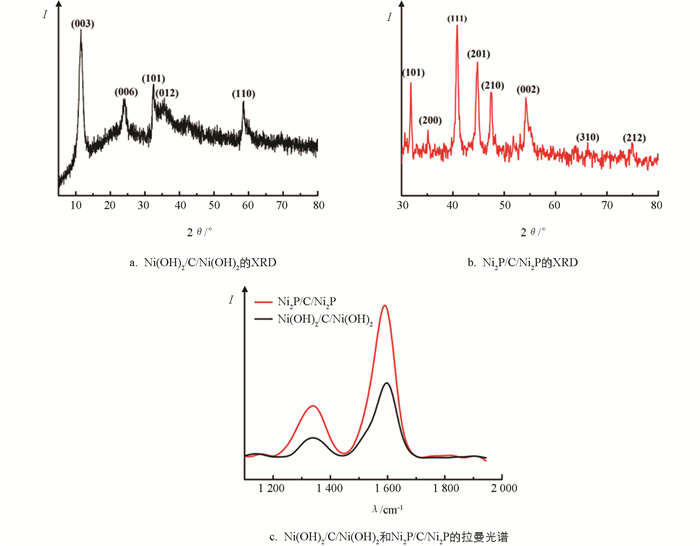
采用X射线衍射(XRD)对制备的材料进行晶体结构分析. 图 1a为在介孔碳球上生长氢氧化物的XRD图,所有的衍射峰与α-Ni(OH)2(JCPDS 38-715)的衍射峰相吻合,其位于12.0°,24.2°,33.0°,35.6°和58.4°的衍射峰分别与α-Ni(OH)2的(003)、(006)、(101)、(012)、(110)晶面相对应. 图 1b显示了Ni(OH)2/C/Ni(OH)2被磷化后的产物的XRD图,其衍射峰与Ni2P(JCPDS no.04-003-1863)的标准衍射峰一致,位于31.8°,35.2°,40.6°,44.8°,47.4°,54.4°,66.2°和74.8°的峰分别与Ni2P的(101)、(200)、(111)、(201)、(210)、(002)、(310)、(212)晶面相对应. 此外,Ni(OH)2/C/Ni(OH)2和Ni2P/C/Ni2P的XRD图没有显示出其他的衍射峰,说明制备的样品不含杂质,具有较高的纯度.
利用拉曼光谱对HMCSs中碳的结构进行测定. 如图 1c所示,Ni(OH)2/C/Ni(OH)2和Ni2P/C/Ni2P都在波数为1 338,1 591 cm-1处展现出相似的特征峰,其分别与碳材料的D峰和G峰相对应. ID/IG(D峰强度与G峰强度的比值)反映碳的结晶程度[11]. 其ID/IG<1,说明碳的结晶度较好.
-
如图 2a所示,SiO2@C具有实心的球形结构,表面光滑,且大小均一,其尺寸约300 nm. 当通过氢氧化钠溶液去除SiO2后,HMCSs呈空心的球形,且结构保持完好(图 2b). 从图 2c中,可以观察到Ni(OH)2纳米片均匀地生长在HMCSs的表面. 与NaH2PO2发生磷化反应后,HMCSs的表面变得粗糙,且附有颗粒状物质(图 2d). 此外,从少许破裂的HMCSs可以看到,其内腔也包含纳米粒子. 该反应的机理是:HMCSs的表面存在介孔,反应物能自由地扩散到HMCSs的内腔,经过溶剂热反应,Ni(OH)2纳米片均匀地固载在HMCSs的内腔及其表面,形成具有核壳结构的Ni(OH)2/C/Ni(OH)2[12];接下来,经过磷化反应,Ni(OH)2纳米片转变为Ni2P纳米粒子,从而形成具有核壳结构的Ni2P/C/Ni2P.
Ni2P/C/Ni2P的透射电镜(TEM)图像(图 3)显示HMCSs形貌保持完整,Ni2P纳米粒子生长在HMCSs的表面及其内腔上,解决了Ni2P纳米粒子团聚的问题,且有利于缓解体积膨胀的问题.
-
图 4a为Ni2P/C/Ni2P电极在扫描速率为0.5 mV/s、电压范围为0.005~3.0 V下测定的CV曲线. 在首次放电过程中,其还原峰位于0.71 V,该峰的形成与Li+嵌入Ni2P以及形成固体电解质中间相(SEI)膜有关,其具体过程为:Ni2P+3Li++3e-→Li3P+2Ni[13]. 在首次放电后,第2圈的还原峰移至1.43 V,第3圈至第5圈的还原峰移至1.57 V,该现象由SEI膜效应引起[14]. 在充电过程中,其氧化峰一直位于1.25 V,该峰的形成归结于SEI膜的分解和Li3P去锂化过程,其具体反应机理为:Li3P+2Ni→Ni2P+3Li++3e-[15].
-
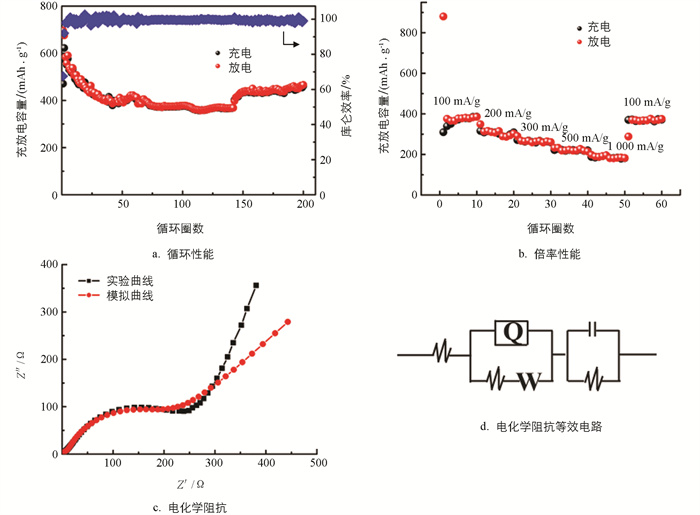
Ni2P/C/Ni2P电极在电流密度为100 mA/g、电压范围为0.005~3.0 V下的充放电曲线如4b所示,在首次放电过程中,电压平台位于1.35 V,其后无明显的电压平台;在充电过程中,无明显的电压平台出现. Ni2P/C/Ni2P的初始放电容量和充电容量分别为696、470 mAh/g,容量保留率约为67.5%,其容量衰减是由SEI膜的形成和电解质的分解导致的[16]. 但在随后的充放电循环中,库仑效率上升至99%以上. 此外,第200圈循环时的充放电容量比第50,100圈循环时的容量高,这是由于电极材料的活化以及电解液分解生成聚合物/凝胶膜的缘故[17]. 该聚合物/凝胶膜可以缓冲充放电过程中产生的体积膨胀,并能在低电位下传送额外的容量,从而进一步提高了Ni2P/C/Ni2P的循环性能.
-
图 5a为Ni2P/C/Ni2P在电流密度为100 mA/g下的循环性能曲线. Ni2P/C/Ni2P电极循环200圈后,其放电容量保持在465 mAh/g,库仑效率接近100%. 此外,当Ni2P/C/Ni2P循环到140圈后,其容量上升,这是由于电极材料的活化以及电解液分解生成聚合物/凝胶膜的缘故.
-
图 5b为Ni2P/C/Ni2P电极在不同电流密度下的倍率性能. 在电流密度为100,200,300,500,1 000 mA/g下,Ni2P/C/Ni2P的放电容量分别为~385,~294,~264,~218,~185 mAh/g. 当电流再次设置为100 mA/g时,其放电比容量恢复到375 mAh/g. 结果表明,Ni2P/C/Ni2P具有良好的倍率性能.
-
为了研究电极材料的导电性,本实验对Ni2P/C/Ni2P电极在频率0.1~10 000 Hz范围内进行了电化学阻抗(EIS)测试. 实验及模拟尼奎斯特图如图 5c所示,尼奎斯特图由高频区域的半圆形和低频区的斜线组成. 半圆的直径与锂离子电荷转移电阻(Rct)和Li+在SEI膜上的传输阻抗(Rf)有关,斜线的斜率与锂离子在固体中的扩散速度(Zw)有关[18]. 根据模拟等效电路图(5d),Ni2P/C/Ni2P的Rct值为160.9 Ω,其导电性良好.
2.1. X射线衍射(XRD)和拉曼光谱分析
2.2. 形貌分析
2.3. 电化学性能分析
2.3.1. 循环伏安性能曲线
2.3.2. 充放电曲线
2.3.3. 循环性能曲线
2.3.4. 倍率性能曲线
2.3.5. 电化学阻抗图谱
-
本研究以空心的介孔碳球(HMCSs)为载体,在其空腔内部及表面固载Ni(OH)2纳米片,接下来通过低温磷化过程,合成具有核壳结构的Ni2P/C/Ni2P复合物. 该微观结构一方面具有大的比表面积,使电极材料与电解液的接触面积增大,有利于锂离子的运输,另一方面其内部具有大的空腔,能够缓解其体积膨胀. 此外,空心碳球能改善电极材料的导电性,从而提高其储锂性能. 因此,Ni2P/C/Ni2P被作为锂离子电池负极材料,在电流密度100 mA/g下,循环200圈后,其容量保持在465 mAh/g. 该方法可用于其他磷化物及电极材料的设计与合成.








 DownLoad:
DownLoad: