-
开放科学(资源服务)标识码(OSID):

-
柑橘作为世界第一大类水果,在国际贸易中占据重要地位[1]。柑橘黄龙病(Citrus Huanglongbing,HLB),又称“黄梢病(Yellow shoot)”“青果病(Greening)”,由韧皮部杆菌属亚洲种(Candidatus Liberibacter asiaticus,CLas)引起,该病菌侵染诱导韧皮部组织缓慢且系统性的免疫反应,包括防卫相关基因的表达、胼胝质沉积[2],是一种对柑橘产业具有毁灭性的病害[3-4],严重威胁我国柑橘产业的可持续发展[5]。柑橘感染黄龙病后,叶片呈现黄绿斑驳、畸形,果实变小、畸形、品质下降,根系腐烂,最终全株枯死[6]。HLB主要为害芸香科的柑橘属(Citrus)和枳属(Poncirus)植物[7],能通过柑橘木虱、带病苗木嫁接及菟丝子(Cuscuta)传播。最新研究表明,黄龙病显症叶片中CLas的增殖会显著降低内生菌群落多样性[8],因此通过深入挖掘柑橘内生细菌资源,筛选具有生防潜力的功能菌株对于黄龙病的防控具有重要意义。柑橘黄龙病作为一种毁灭性病害,至今仍缺乏有效抗病品种与根治药剂,目前主要依靠培育无毒苗木、严格砍除病树、大面积防治木虱进行综合防控[9-14]。抗生素处理[15-16]和生物防治[17-18]也有报道,但前者有悖于绿色防治的植保方向,后者只能作为辅助手段。目前,许多学者对柑橘内生细菌分离进行了研究,Lacava等[19]从柑橘中分离出一株能够抑制柑橘传染性色变病原菌繁殖的内生细菌;刘起丽等[20]分离筛选出5株对柑橘青霉病菌有明显拮抗作用的内生细菌;Munir等[21]从柑橘中分离出一株枯草芽胞杆菌(Bacillus subtilis)L1-21,研究表明其对CLas具有一定拮抗作用,在感病果园中病叶中脉的CLas滴度从109 log CFU/g下降到104 log CFU/g;Yang等[22]利用多黏芽孢杆菌(Paenibacillus polymyx)KN-03对感染CLas的柑橘植株进行24次灌根处理后,柑橘植株内CLas滴度显著降低,且促进了柑橘叶片的生长以及刺激活性氧的产生。这些内生菌的研究与应用为黄龙病的防控提供了新方向。由于Clas至今仍无法实现体外人工培养,其致病机理研究与防治技术研发受到了极大的制约,目前尚未发现有效防控黄龙病的抗病品种或特效药剂。本研究从重病柑橘果园无黄龙病症状植株组织中进行内生细菌分离,对内生细菌进行组织抗性和盆栽抗性评价,筛选出优良生防菌株,再对筛选出的生防菌株进行鉴定。
HTML
-
试验于2023-2024年在西南大学柑桔研究所国家苗木脱毒中心实验室完成。
植株:实验室前期芽接接种CLas的粗柠檬(Citrus jambhiri)保存于本实验室隔离温室中;自广西、江西等地重病柑橘园采集的无症植株样品。
NA培养基(Nutrient Agar Medium):蛋白胨10 g/L,牛肉浸粉5 g/L,葡萄糖10 g/L,琼脂15 g/L,使用NaOH调节pH值为7.0,121 ℃高温灭菌20 min,4 ℃冰箱保存。
-
本研究使用的引物名称、序列及用途见表 1。
-
自广西、江西等地重病柑橘园采集无症植株样品,采用常规的组织分离方法分离内生细菌。用无菌水冲洗柑橘无症状叶片,在无菌的操作环境下,将其放置在干燥的无菌滤纸上晾干,使用无菌刀片将叶片切取大小约为1.0 cm×1.0 cm的叶片组织和长度为1.0 cm的叶脉组织。将切取的叶片和叶脉组织置于浓度为75%的酒精中消毒浸泡约30 s,再将叶片和叶脉组织置于装有3%次氯酸钠的烧杯中浸泡1 min,接着再次把叶片和叶脉组织置于浓度为75%的酒精中浸泡约30 s,使用无菌水将其冲洗3次后,用干燥的无菌滤纸吸干水分;用酒精灯灼烧过的镊子挑取叶片和叶脉组织接种至灭菌后NA固体培养基上,每个平板各整齐放置4块叶片和叶脉组织,在28 ℃条件下的恒温恒湿培养箱中倒置培养2~3 d,观察统计菌落生长状况。
-
由于黄龙病菌无法体外培养,采用两种指示病原菌平板对峙试验的方法,对黄龙病菌的潜力生防菌株进行初步筛选。将指示病原菌菌株Pst DC3000、Rst GMI100培养至对数生长期,采用梯度稀释法调整浓度为107 CFU/mL和108 CFU/mL,分别用无菌拭子均匀涂抹在NA固体培养基上晾干。使用无菌打孔器将滤纸片打成直径为6.0 mm大小的小型滤纸片,再用无菌镊子将小型滤纸片分别置于固体培养基两侧,滴加内生细菌菌悬液(OD600=0.8)至滤纸片上晾干。试验设置阳性对照(34 mg/mL四环素)和阴性对照(无菌水),重复3次试验,培养48 h后观察抑菌圈并测量其直径。
-
参考谢帆[23]的方法并有所改动,采用半叶法对潜力生防细菌的组织拮抗活性进行评价。将感染黄龙病且大小相同、生长状态相似的叶片用无菌水清洗干净,放置在无菌培养皿中,叶柄处包裹微湿不滴水的无菌棉,培养皿中垫上2片滤纸并滴入适量无菌水以保湿。将生防细菌菌液摇至OD600=0.8后,在叶片背面未取样的平行一侧进行针刺处理(用注射器的针在叶片上轻刺3~7下,注意不要戳破叶片),并用注射器注射相同量(0.5 mL)的菌液,分别做内生菌处理、抗生素(盐酸四环素)处理和水处理,试验设置4个重复。用无菌滤纸擦干后密封培养皿,置于28 ℃恒温培养箱中培养96 h。培养结束后,从针刺位置附近取样,提取DNA并进行qPCR定量检测,比较生防菌处理前后黄龙病菌含量的变化,通过数据分析和作图展示试验结果。
-
将PCR检测黄龙病阳性的叶片根据形状大小、生长状态相似性均匀分组,分别提取阳性叶片在潜力生防细菌处理前和处理后96 h的DNA,定量到200 ng/μL。以HLB-qF/HLB-qR为引物,以柑橘COX基因为内参基因(CsCOX-F/CsCOX-R)进行qPCR检测。试验设置3个生物学重复,3个技术性重复,采用2-ΔΔCT法计算CLas的相对含量(引物序列详见表 1)。以CLas相对含量的数值来评价潜力生防细菌的组织抑菌活性。使用Student's t-test或ANOVA进行假设检验,分析其显著性,GraphPad Prism 9.0进行绘图(显著水平:p<0.05表示差异显著,p<0.01表示差异极显著)。
-
参考南京[24]和王立等[25]的方法并有所改动。将前期筛选保存的具有显著拮抗作用的生防菌株接种于NA液体培养基中,37 ℃培养至生长对数期。试验设置内生菌处理、抗生素(盐酸四环素)处理和无菌NA培养基处理,各6个生物学重复。选取发病一个月左右、叶片有明显症状的粗柠檬植株,每株植株浇灌200 mL菌悬液,每周浇灌1次,共浇灌8次。分别于0 dpi(days post infection,dpi)、30 dpi、60 dpi、90 dpi按植株东、南、西和北方位定量采集叶片样品,分离叶脉组织液氮速冻后,转移至-80 ℃冰箱。一部分样品使用植物DNA基因组提取试剂盒提取DNA,用于qPCR检测;其余样品供后续进行相关拮抗活性评价测定使用。
CLas的实时定量检测方法同1.6。使用植物胼胝质ELISA检测试剂盒定量测定样品中胼胝质含量,具体步骤参考说明书。
-
根据《伯杰氏细菌手册(第八版)》和《常见细菌系统鉴定手册》进行形态学鉴定、革兰氏染色及生理生化指标测定[26]。使用细菌DNA基因组提取试剂盒提取潜力生防细菌DNA后,采用细菌16S通用引物27F和1492R扩增16S rRNA序列,将送至擎科公司进行测序的PCR产物基因序列进行BLAST序列比对,下载同源性较高的序列,再利用MEGA 11.0软件采用邻接法(Neighbor-Joining)构建系统发育树,确定潜力生防菌株的分类地位。
-
将生防细菌种子液按1%接种量接入100 mL NA培养基(250 mL锥形瓶),于37 ℃、180 r/min摇床培养。接种后开始监测,每隔1 h监测一次,监测时间范围为0 h到24 h。测定吸光度时,以无菌LB液体培养基作为空白对照,将分光光度计的波长设置为600 nm,使用移液器将菌液缓慢加入比色皿中,注意避免产生气泡,每个样品重复测定3次,以获取不同时间点的吸光度数据,生长曲线使用GraphPad Prism 9.0进行绘制。
1.1. 供试材料
1.2. 引物设计
1.3. 柑橘内生细菌的分离、纯化
1.4. 平板对峙试验
1.5. 潜力生防细菌的组织抗性评价
1.6. CLas的实时定量检测
1.7. 盆栽试验
1.8. 潜力生防菌株的鉴定
1.9. 生防细菌的生长曲线
-
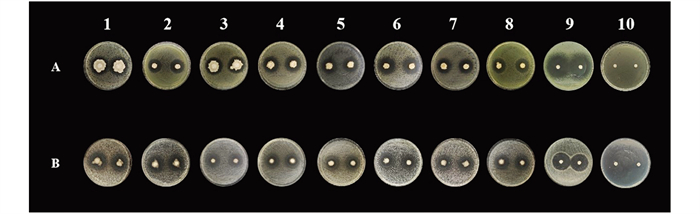
采用传统的组织分离法从发病严重的果园中采集无症状植株叶片,从中分离和纯化可培养的内生细菌63株。通过平板对峙法评估,共筛选出13株对Pst DC3000和Rst GMI100均有拮抗作用的内生细菌,其中,NS12Y101对Pst DC3000和Rst GMI100的抑菌圈最大,均大于30 mm。包括NS18Y101在内的7个菌株对2种指示病原菌的抑菌圈直径均大于20 mm(表 2、图 1),表明其具有较高的抗菌活性;其余5株菌株也表现出一定的抗菌活性。
-
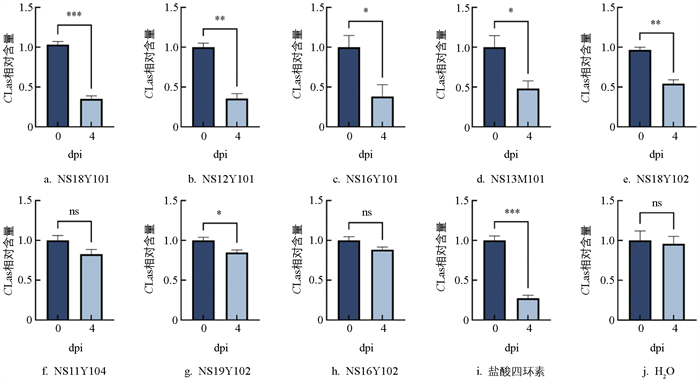
对前述8株潜力生防菌株利用半叶法进行了组织抗性评价。qPCR分析结果显示,在试验处理后96 h,CLas相对含量在阳性对照(抗生素处理组)中极显著下降,在水对照组中无显著变化,说明试验数据可靠。所有潜力生防菌株处理组中CLas相对含量均表现为下降,其中NS18Y101菌株处理组CLas相对含量降幅最大,达到极显著水平(p<0.001);NS12Y101、NS18Y102组表现为显著下降(p<0.01);NS16Y101、NS13M101和NS19Y102菌株处理组也呈现显著下降(p<0.05);NS11Y104、NS16Y102菌株处理组无显著差异(图 2)。试验结果初步表明NS18Y101对CLas具有良好的抑菌效果。
-
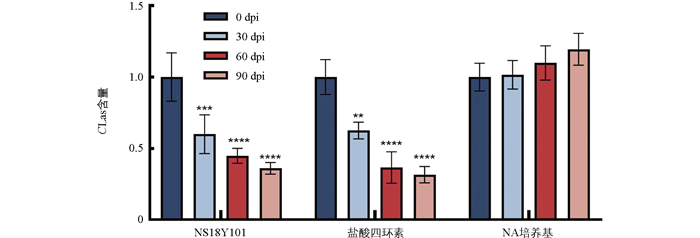
为了进一步评估NS18Y101对CLas的拮抗效果,以盐酸四环素为阳性对照,以NA培养基为阴性对照,每处理组设置6个生物学重复开展盆栽拮抗评价。CLas的qPCR检测结果如图 3所示,与0 dpi(days post inoculation)相比,在30 dpi、60 dpi和90 dpi时,NS18Y101处理植株中的CLas相对含量均极显著下降,与同期抗生素处理植株的结果相似,而NA培养基处理植株中CLas相对含量无显著变化,呈逐渐小幅上升趋势。这进一步说明NS18Y101对CLas具有良好的拮抗作用。
-
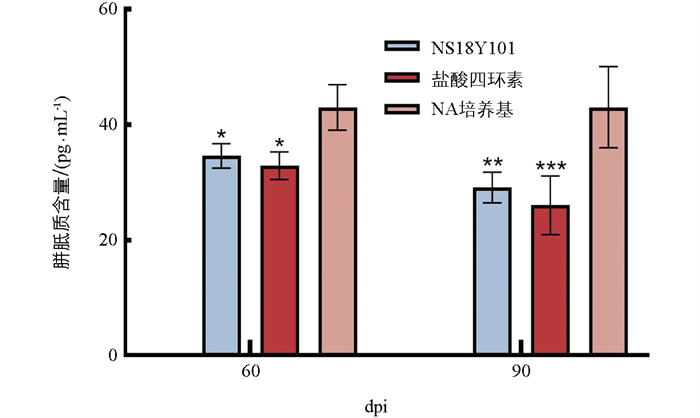
在生防菌株处理60 dpi和90 dpi后,对柑橘植株进行胼胝质的测定。结果显示,与对照NA培养基处理相比,NS18Y101处理后胼胝质含量均显著降低,与抗生素处理相当(图 4)。这表明NS18Y101可显著降低CLas侵染所导致的胼胝质积累。
-
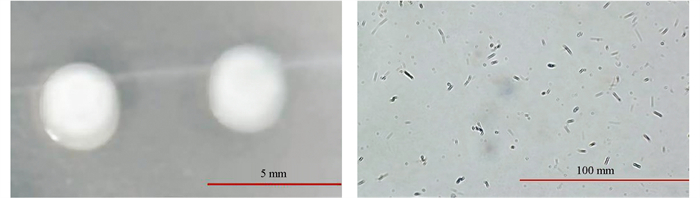
在体式显微镜下观察到NS18Y101的菌落为表面有光泽,边缘平滑且凸起白色圆形;其细胞形状为杆状,孢子为圆形或椭圆形;革兰氏染色结果表明NS18Y101为革兰氏阳性菌(图 5)。
-
根据《常见细菌系统鉴定手册》对NS18Y101进行了部分生理生化测定,结果见表 3。生理生化特征分析显示,菌株NS18Y101具有硝酸盐还原的能力,且在V-P、MR及吲哚试验中均呈阴性反应,表明其缺乏葡萄糖发酵能力及色氨酸分解代谢途径。该生防菌株还可高效分解葡萄糖、蔗糖、D-甘露醇及D-山梨醇等碳水化合物,但缺乏乳糖代谢能力。氮源利用方面,NS18Y101菌株可利用硝酸铵、硫酸铵、氯化铵及尿素等无机氮源。环境适应性评估显示,NS18Y101生防菌株可达到7%的耐盐阈值,且可在60 ℃高温环境中存活,但在低温(4 ℃)条件下均无法存活。酸碱耐受性试验表明,杆菌NS18Y101可在pH值5.7~9.0范围内生长。
-
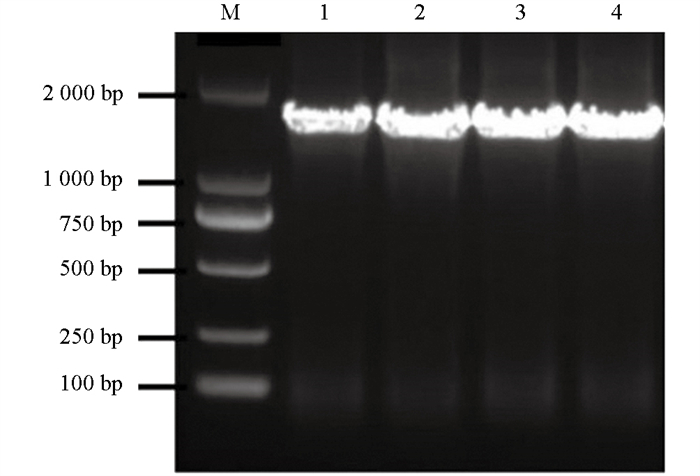
提取NS18Y101菌株的DNA,利用细菌16S rRNA基因的通用扩增引物27F/1492R进行PCR。结果显示成功扩增出单一且清晰的条带,与预期长度一致(图 6),测序用于进一步序列分析。
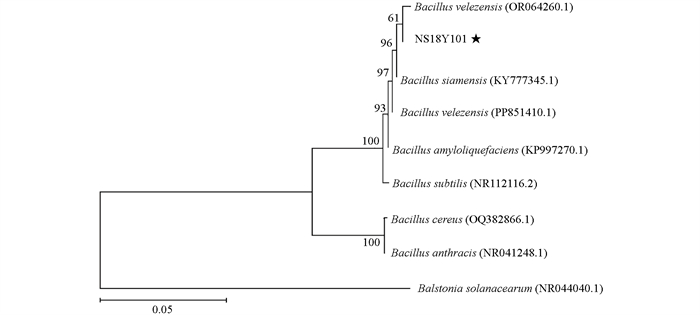
将NS18Y101的16S rRNA序列在NCBI数据库BLAST中进行BLAST比对,下载同源性较高的序列并构建系统发育树。序列比对结果显示NS18Y101与Bacillus velezensis序列同源性最高,为100 %。在系统发育树上,NS18Y101与Bacillus velezensis(OR064260.1)聚为一枝,形态特征、生理生化特征结合分子序列测定结果确定NS18Y101为贝莱斯芽孢杆菌(图 7)。
-
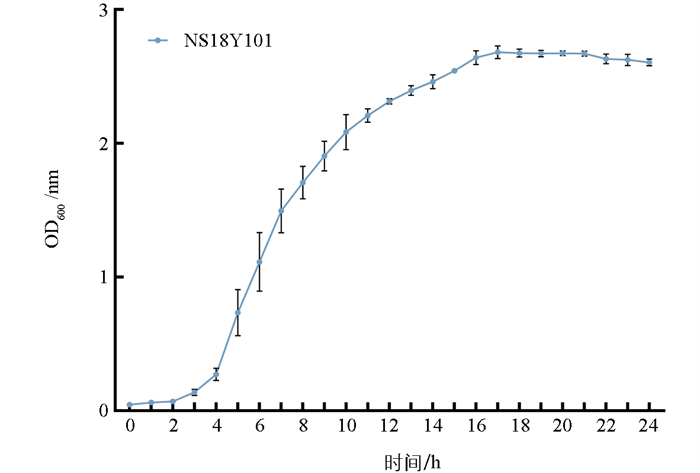
对具有CLas显著拮抗作用的菌株NS18Y101进行生长曲线测定。试验结果如图 8显示,菌株在0~3 h为迟缓期,OD600值增长缓慢,接着进入对数期,4~16 h期间芽孢杆菌迅速繁殖,OD600值急剧上升。16~21 h进入稳定期,生长速度相对稳定,OD600值均在2.700上下波动。21 h后进入衰亡期,OD600值逐渐下降。
2.1. 潜力生防细菌的离体抗性评价
2.2. 潜力生防细菌的组织抗性评价
2.3. 生防细菌的盆栽拮抗评价
2.3.1. 生防菌株处理对CLas相对含量的影响
2.3.2. 生防菌株处理对植株胼胝质含量的影响
2.4. NS18Y101的鉴定
2.4.1. NS18Y101的形态学鉴定
2.4.2. NS18Y101的生理生化鉴定
2.4.3. NS18Y101的分子生物学鉴定
2.5. NS18Y101的生长曲线测定结果
-
作为全球第一大经济作物,柑橘产业已在我国覆盖20余个省份,其年产量和种植面积均居世界前列[27]。然而,由韧皮部杆菌引发的柑橘黄龙病已成为威胁柑橘产业可持续发展的毁灭性病害,具有发病迅速、防治困难、致死率高等特点,可导致染病植株在3~5年内完全丧失结果能力,造成重大经济损失[28]。目前,针对柑橘黄龙病的防治手段仍以化学药剂和砍伐病树为主,但长期使用农药易导致环境污染和抗药性问题,且无法从根本上解决病原菌的系统性侵染难题。已有不少学者研究发现抗生素在柑橘黄龙病防治中具有显著效果,赵学源等[14]研究发现,带病芽条经过四环素族抗生素浸泡后嫁接、生长正常的植株在5~6年内没有复发病害,亦不带有病原;姚廷山[15]等研究发现土霉素可有效抑制黄龙病菌亚洲种,减少柑橘叶片内淀粉含量,增加柑橘产量。因此,本试验通过组织抗性评价和盆栽拮抗活性评价对前期筛选的8株对指示病原菌具有明显拮抗作用的潜力生防细菌开展研究,其中贝莱斯芽孢杆菌NS18Y101能够显著抑制CLas的增长,抑制效果与抗生素相当,可降低植株中胼胝质的积累,表明这株生防细菌菌株具备开发为柑橘黄龙病生物防治菌剂的潜力。但是经过贝莱斯芽孢杆菌NS18Y101处理的黄龙病植株与对照组相比症状并没有减轻,可能是由于黄龙病植株的免疫力会随病害的侵染逐渐下降,若植株已处于衰弱状态(筛管坏死),即使CLas被抑制,植株也难以恢复正常生长,症状因此无法缓解。
目前,贝莱斯芽孢杆菌NS18Y101可能是通过产生抗菌肽、脂肽类化合物等直接破坏CLas的细胞膜结构,抑制其DNA复制和代谢活动,从而降低病原菌活性;也可能与CLas争夺柑橘树体内的碳源、氮源及铁离子等关键营养物质,抢占生态位,从而抑制CLas增殖;亦或是激发柑橘树自身防御系统,提升过氧化物酶、多酚氧化酶等防御酶活性,增强植株对CLas的抗性,来减少病原菌侵染和扩散。具体的作用机制目前尚不清楚,还有待挖掘。
-
有研究发现,贝莱斯芽孢杆菌在抑制CLas具有重要作用,如胡党振等[29]所报道的贝莱斯芽孢杆菌No.4菌剂可改善根际环境,从而诱导柑橘植株的抗黄龙病能力[29]。为提高生防菌株的抑菌潜力和生态适应性,本研究从发病严重的生产果园中无症状植株上采集了叶片样本,用于分离、筛选出对CLas具有高效抑菌活性的生防细菌。由于黄龙病菌无法体外培养,本研究以丁香假单孢杆菌、番茄劳尔氏菌为指示病原菌,通过对叶片样品中的微生物进行挖掘,从叶片样本片中分离得到13株具有生防潜力的内生细菌;又通过组织抗性评价和盆栽评估法筛选出对CLas具有显著拮抗作用的NS18Y101;再经过形态学、分子生物学、生理生化鉴定,发现NS18Y101为贝莱斯芽孢杆菌菌株。本研究成功分离并筛选出1株具有生防潜力、稳定代谢活性和环境适应能力的芽孢杆菌,为已开展的温室柑橘黄龙病植株防控效果验证提供了重要理论支撑。
-
经过形态学、分子生物学及生理生化综合鉴定,从重病果园无HLB症状植株上分离获得的菌株NS18Y101是对CLas有显著拮抗作用的贝莱斯芽孢杆菌。





 DownLoad:
DownLoad: