-
开放科学(资源服务)标识码(OSID):

-
畜牧业中抗生素的长期使用会导致动物耐药性的增加以及药物残留等问题,对环境和人类健康造成巨大的潜在威胁[1]。以促进动物机体肠道健康发育和提高免疫力为主要功能的益生菌和酶制剂,作为抗生素的替代品为无抗饲料的生产提供了可行方案。益生菌可以有效抑制宿主体内的病原菌,提高宿主的免疫力、增加体质量、提高生产性能、平衡肠道的微生物群[2]。酶制剂是饲料中最重要和最常见的添加剂,可以作为动物内源性酶的有益补充,辅助其降解饲料,促进营养成分的吸收。
益生菌在动物机体内可以分泌产生细菌素、溶菌酶、过氧化氢、蛋白酶等有杀菌或抑菌作用的物质,直接对病原体产生拮抗作用。益生菌产生的有机酸和挥发性脂肪酸(如乳酸、乙酸、丁酸和丙酸)能改变肠道pH值,通过降低肠道pH值抑制病原体在消化道中的定植,从而竞争性地抑制病原体[3];同时益生菌可通过竞争附着位点来阻止病原体在肠道表面的生长,阻碍病原微生物的竞争和结合[4];益生菌还可以平衡肠道微生物,改善肠道完整性,调节免疫功能[5]。粪肠球菌(Enterococcus faecalis)作为常用益生菌常常被添加在鸡的饲粮中。Shehata等[6]研究发现肉鸡口服粪肠球菌1×108 CFU/d,可显著提高肉鸡的饲料转化率。
动物饲粮含有大量非淀粉多糖(NSP),会导致消化物黏度增加,从而抑制动物的生长性能,而酶制剂能降低食糜的黏度。碳水化合物酶能够将NSP水解成更小的低聚糖,使消化液黏度降低并释放营养物质[7]。NSP的水解产物通过益生菌的发酵,可以产生短链脂肪酸(SCFA),而SCFA的浓度增加通常与有益菌数量的增加和病原菌数量的减少相关[8]。纤维素酶是由内切葡聚糖酶、纤维二糖水解酶和β-葡萄糖苷酶组成的一组水解酶[9]。对于非草食动物而言,无法分解细胞壁中的纤维素和果胶等物质,因此在饲料中添加纤维素酶、果胶酶等酶制剂可以帮助鸡分解植物细胞壁,提高饲粮中营养成分的利用率[10]。
目前,关于益生菌或酶制剂的研究和应用在肉鸡和产蛋期的蛋鸡较多,对于酶菌协同添加以及有关0~8周龄蛋鸡的研究相对较少。0~8周龄蛋鸡的免疫和消化器官尚未发育完全,免疫能力较低,且无法分泌足够的内源酶来供机体消化和吸收饲料中的营养成分[11],因此,本研究以0~8周龄蛋鸡为试验对象,探究饲粮中粪肠球菌和酶制剂协同添加对其体质量、体尺、生长激素、抗氧化能力和肠道发育等的影响,为蛋鸡的无抗饲料研究与应用提供参考。
HTML
-
粪肠球菌与复合酶制剂来自挑战集团天津博菲德科技有限公司。粪肠球菌(优C-100):活菌数≥1×1010 CFU/g;复合酶制剂(8308)主要成分:蛋白酶≥12 000 U/g、淀粉酶≥5 000 U/g、β-葡聚糖酶≥2 500 U/g、木聚糖酶≥30 000 U/g、纤维素酶≥500 U/g。蛋鸡基础饲粮为正大521蛋小鸡配合饲料,主要营养成分(%):粗蛋白≥20.0,水分≤14.0,钙0.6~1.2,总磷0.5,氯化钠0.2~0.8,粗纤维≤6.0,粗灰分≤8.0,蛋氨酸+胱氨酸≥0.74。
-
采用单因素随机试验设计,试验动物选择体质量相近的120只1日龄海兰褐蛋鸡(雌性),随机分为4个组,每组5个重复,每个重复6只鸡。对照组(Con组)饲喂基础饲粮,复合酶组(CE组)在基础饲粮中添加酶制剂100 mg/kg,粪肠球菌组(EF组)在基础饲粮中添加粪肠球菌100 mg/kg,酶菌协同组(CE+EF组)在基础饲粮中添加酶制剂与粪肠球菌各100 mg/kg,试验期为8周。试验鸡群采用自由采食、自由饮水的方式进行饲养,并按照育雏期免疫程序对鸡群进行免疫处理。
-
测定血液抗氧化指标及生长激素指标试剂盒:超氧化物歧化酶(SOD)、谷胱甘肽过氧化物酶(GPX)、总抗氧化能力(T-AOC)、生长激素(GH)、生长激素受体(GHR)均来自上海酶联生物。
-
试验第8周结束后,海兰褐蛋鸡翅下静脉采血10 mL并分装到不含抗凝剂的采血管中,静置15 min后,以3 500 r/min离心15 min取上层血清分装到冻存管中置于-20 ℃冰箱和液氮速冻后置于-80 ℃冰箱中备用。对鸡进行动脉放血处死后,解剖分离十二指肠、空肠、回肠,将各肠段剖开清理内容物,取十二指肠、空肠、回肠中段各1 cm,置于4%的多聚甲醛中固定保存,同时取盲肠内容物分装于冻存管中,液氮速冻后置于-80 ℃冰箱中备用(西南大学实验动物伦理审查委员会批准,伦理审查号:LAC2025-2-0127)。
-
体尺指标按照《家禽生产性能名词术语与度量计算方法》 (NY/T 823—2020)测定。用微量法测定抗氧化指标SOD、T-AOC、GPX,使用酶联免疫分析法(ELISA)测定GH、GHR的含量,具体检测方法按试剂盒说明书进行。十二指肠、空肠、回肠组织切片制作完成后,通过显微成像系统进行拍照,随机挑选10个完整的肠绒毛以及隐窝利用Image软件进行测量,并计算绒隐比(绒毛高度/隐窝深度)。
-
数据使用SPSS 2022进行ANOVA单因素方差分析,并用Duncan法进行多重比较,LEfse差异分析阈值,p<0.05为差异有统计学意义。盲肠16 S rDNA测序由杭州联川生物技术股份有限公司完成;以p<0.05,LDA>4来筛选不同组间的差异微生物;使用R包进行相关性分析,选择盲肠菌群属水平中相对丰度前20的菌群与表型数据进行Spearman相关性分析。
1.1. 试验材料
1.2. 试验动物与设计
1.3. 测定试剂盒
1.4. 样品采集
1.5. 试验方法
1.6. 数据分析
-
由表 1可知,在饲粮中添加粪肠球菌(EF)和复合酶制剂(CE)对0~8周龄蛋鸡的体斜长、胸深、胫围、胫长均无明显影响(p>0.05),但CE+EF组相比于Con组有提高体斜长、胸深、胫围和胫长的趋势。CE+EF组、CE组显著提高了0~8周龄蛋鸡的龙骨长(p<0.05);另外,体质量指标CE+EF组显著高于Con组(p<0.05);胸宽指标CE+EF组显著高于CE组和Con组(p<0.05)。
-
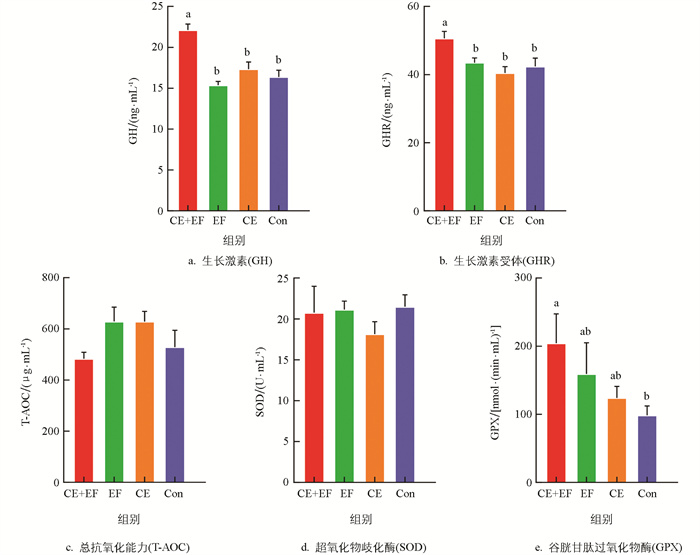
由图 1可知,对于血清中GH和GHR含量,CE+EF组显著高于EF组、CE组和Con组(p<0.05),EF组、CE组、Con组间差异无统计学意义(p>0.05)。对于抗氧化指标方面,无论是单独添加复合酶制剂(CE)、粪肠球菌(EF),还是作为酶菌协同添加(CE+EF组)对0~8周龄蛋鸡的总抗氧化能力(T-AOC)、超氧化物歧化酶(SOD)活性都无显著影响(p>0.05);对于谷胱甘肽过氧化物酶(GPX)活性,CE+EF组显著高于Con组(p<0.05);EF组、CE组与Con组间差异无统计学意义(p>0.05),但EF组和CE组相比于Con组有提升的趋势。
-
由表 2可知,在十二指肠中,CE+EF组的绒毛高度显著高于EF组、CE组和Con组(p<0.05),EF组绒毛高度显著低于Con组(p<0.05);EF组和CE组的隐窝深度均显著低于CE+EF组和Con组(p<0.05);绒隐比差异无统计学意义(p>0.05)。对于空肠,在绒毛高度方面,EF组显著高于CE组(p<0.05);隐窝深度方面,CE组会增加隐窝深度,与CE+EF组、EF组、Con组间差异有统计学意义(p<0.05);绒隐比方面,EF组最大,显著高于CE+EF组、CE组、Con组(p<0.05),CE组绒隐比最小,显著低于其他3组(p<0.05),CE+EF组与Con组间差异无统计学意义(p>0.05)。对于回肠,Con组的绒毛高度显著低于CE+EF组、EF组和CE组(p<0.05);隐窝深度各组间差异无统计学意义(p>0.05);绒隐比方面,Con组最小,显著低于CE+EF组、EF组(p<0.05)。
-
由表 3可知,与Con组相比,EF组Shannon指数、Simpson指数显著降低(p<0.05),其余组间差异无统计学意义(p>0.05)。对于Chao1指数、ACE指数,4组间差异均无统计学意义(p>0.05)。
-
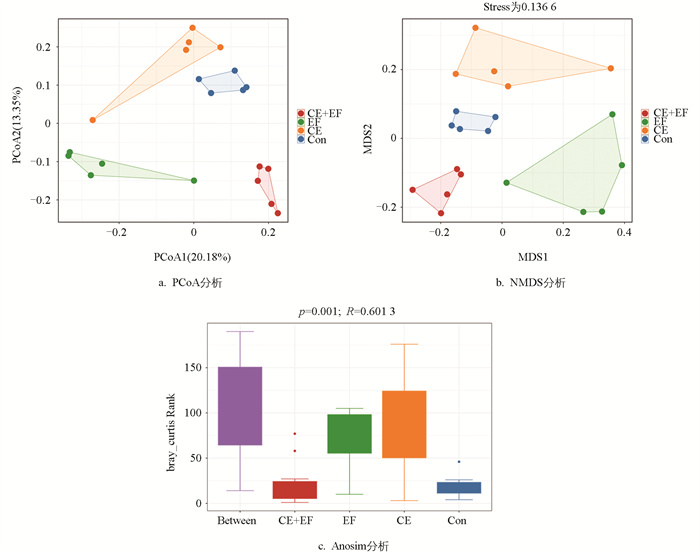
基于Bray-Curtis距离的主坐标分析对不同饲粮组盲肠微生物进行Beta多样性的降维分析。PCoA分析显示,CE+EF组、EF组、CE组和Con组内存在较好的聚类,组间分开趋势明显,说明各组之间的盲肠微生物组成存在显著差异(图 2a)。NMDS分析显示,各组的微生物组成也存在显著差异,同时Stress为0.136 6<0.2,证明数据可用(图 2b)。Anosim分析检验后,p=0.001,证明组间差异大于组内差异,试验分组有意义(图 2c)。
-
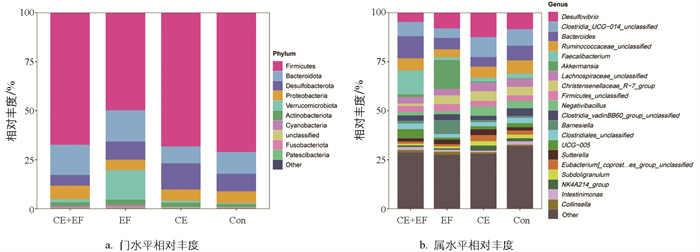
进一步分析不同饲粮组盲肠微生物在门、属水平上的相对丰度,发现门水平上,各试验组主要由厚壁菌门(Firmicutes)、拟杆菌门(Bacteroidota)、脱硫杆菌门(Desulfobacterota)、变形菌门(Proteobacteria)、疣微菌门(Verrucomicrobiota)构成(图 3a)。属水平上,CE+EF组的优势菌群主要为粪杆菌属(Faecalibacterium)、拟杆菌属(Bacteroides)、瘤胃球菌科属(Ruminococcaceae_unclassified)、Clostridia_UCG-014_unclassified、脱硫弧菌属(Desulfovibrio);EF组的优势菌群为阿克曼氏菌属(Akkermansia)、脱硫弧菌属(Desulfovibrio)、巴恩斯氏菌属(Barnesiella)、拟杆菌属(Bacteroides)、Clostridia UCG_014 unclassified;CE组的优势菌群为脱硫弧菌属(Desulfovibrio)、Clostridia_UCG-014_unclassified、拟杆菌属(Bacteroides)、瘤胃球菌科属(Ruminococcaceae_unclassified)、克里斯滕森菌属(Christensenellaceae_R-7_group)、Negativibacillus;Con组的优势菌群为脱硫弧菌属(Desulfovibrio)、Clostridia_UCG-014_unclassified、拟杆菌属(Bacteroides)、瘤胃球菌科属(Ruminococcaceae_unclassified) (图 3b)。CE+EF组相比于CE组、EF组、Con组粪杆菌属的相对丰度增加明显,同时还增加拟杆菌属的相对丰度。
-
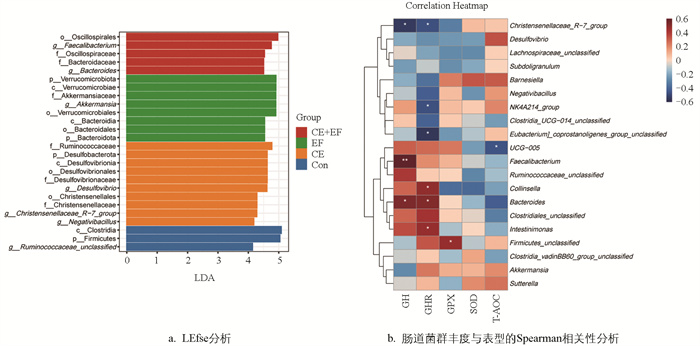
为了寻找各组间的差异微生物,以p<0.05,LDA>4为阈值进行LEfse(LDA Effect Size)差异分析。由图 4a可知,CE+EF组显著富集了粪杆菌属(Faecalibacterium)、拟杆菌科(Bacteroidaceae)、拟杆菌属(Bacteroides)、颤螺菌科(Oscillospiraceae);EF组显著富集了疣微菌门(Verrucomicrobiota)、拟杆菌门(Bacteroidota)、Akkermansiaceae、阿克曼氏菌属(Akkermansia);CE组显著富集了脱硫杆菌门(Desulfobacterota)、脱硫弧菌科(Desulfovibrionaceae)、瘤胃球菌科(Ruminococcaceae)、克里斯滕森科(Christensenellaceae)、脱硫弧菌属(Desulfovibrio)、克里斯滕森菌属(Christensenellaceae_R-7_group)、Negativibacillus;Con组显著富集了厚壁菌门(Firmicutes)和Ruminococcaceae unclassified。
为了进一步分析肠道菌群与表型之间的关系,选择属水平相对丰度前20的菌群与GH、GHR以及抗氧化指标进行Spearman相关性分析。由图 4b可知,GH含量与粪杆菌属(Faecalibacterium)和拟杆菌属(Bacteroides)相对丰度呈显著正相关(p<0.05),Christensenellaceae_R-7_group的相对丰度与GH、GHR含量呈显著负相关(p<0.05);而GHR含量与拟杆菌属(Bacteroides)、科林斯菌属(Collinsella)、Intestinimonas的相对丰度显著正相关(p<0.05);GPX含量与Firmicutes_unclassified相对丰度呈显著正相关(p<0.05)。
2.1. 不同饲粮组对0~8周龄蛋鸡生长发育的影响
2.1.1. 对蛋鸡体质量和体尺指标的影响
2.1.2. 对蛋鸡血液指标的影响
2.2. 对蛋鸡肠道形态的影响
2.3. 不同饲粮组对0~8周龄蛋鸡盲肠微生物的影响
2.3.1. 盲肠微生物Alpha多样性分析
2.3.2. 盲肠微生物Beta多样性分析
2.3.3. 物种组成分析
2.3.4. 肠道微生物差异分析及菌群与表型相关性分析
-
粪肠球菌(Enterococcus faecalis)是兼性厌氧性革兰氏阳性菌,具有强大的环境适应能力,且对多种抗生素具有强耐受性,能够在动物肠道黏膜表层粘附,增强其肠道屏障功能,从而阻隔病原菌接触,并降低侵染机体的概率,是常见且理想的抗生素替代品[12]。另外,粪肠球菌与酶制剂均可以提升、补充动物机体消化酶的活性,促进营养成分(蛋白质、糖类、纤维素等)的分解,促进动物的生长。武震宇等[13]研究发现粪肠球菌能提升育成期籽鹅血清总蛋白和白蛋白的含量,王禹盟[14]对海兰褐雏鸡采用粪肠球菌与粪菌移植处理后,发现添加粪肠球菌可以提高雏鸡的体质量和胫骨长。本研究结果表明粪肠球菌与复合酶制剂协同添加能够显著提高0~8周龄蛋鸡体质量、龙骨长和胸宽;同时,还检测到酶菌协同组(CE+EF组)蛋鸡的GH、GHR和GPX含量显著高于其他组。在家禽中,GH可以调节类固醇生成、细胞增殖和细胞凋亡,还可以调节促黄体素(LH)和胰岛素样生长因子(IGF)的合成[15]。GPX可以通过将还原型谷胱甘肽(GSH)氧化成谷胱甘肽二硫化物(GSSG),从而催化脂质过氧化物和过氧化氢转化为水和相应的脂质醇[16],因此推测对0~8周龄蛋鸡添加粪肠球菌和复合酶制剂可以提高雏鸡血清GPX水平,增强机体的抗氧化能力和降低氧化应激,同时促进其体内GH和GHR的分泌,促进其生长发育。
-
小肠是鸡消化吸收的主要器官,绒毛高度、隐窝深度以及绒隐比反映了肠道消化吸收的能力,当绒毛高度、绒隐比增加,隐窝深度降低时,肠道的营养吸收能力提高。补充益生菌后可以增强蛋鸡的空肠形态,其绒毛表面积(VSA)增加、绒毛高度与隐窝深度、绒毛宽度和绒毛高度比以及隐窝深度显著降低[17]。Mirsalami等[18]发现益生菌处理后增加了肉鸡十二指肠和空肠的绒毛高度,但在回肠中无明显变化。Wang等[19]发现添加复合益生菌,可以显著降低蛋鸡空肠的隐窝深度。本研究中添加粪肠球菌能增加0~8周龄蛋鸡空肠、回肠的绒毛高度和绒隐比,同时降低十二指肠和空肠的隐窝深度,这与前人研究一致。同时,酶菌协同添加会显著增加十二指肠的绒毛高度、回肠的绒毛高度和绒隐比,证明粪肠球菌或者酶菌协同可以增强0~8周龄蛋鸡的肠道形态、维护肠道健康。值得注意的是,与对照组相比,单独添加复合酶制剂能增加回肠的绒毛高度,降低十二指肠的隐窝深度,但会增加空肠的隐窝深度,降低空肠的绒毛高度和绒隐比,出现这种差异的原因还需要进一步探索。
-
肠道菌群与宿主之间的联系非常紧密,肠道菌群可以通过各种途径来影响家禽的生长发育和新陈代谢。Zhang等[20]采用粪便微生物群移植法,用成年鸡(高体质量)肠道内的优势菌群(微杆菌属和鞘氨单胞菌属)重塑1日龄雏鸡的肠道微生物群,从而提高其生长性能,而脂肪代谢与优势菌群的相对丰度呈正相关,说明肠道微生物可以通过调节脂肪代谢提高鸡的生长性能。
在饲粮中添加益生菌会影响鸡的肠道菌群,从而改善鸡的肠道健康等。付文娟等[21]通过饲喂间型脉孢菌培养物,发现该益生菌有助于优化鸡的盲肠菌群结构,并且改善生产性能与健康程度。在本研究中,添加粪肠球菌后增加了拟杆菌门的相对丰度。拟杆菌门的微生物可以产生乙酸、丙酸、甲酸和丁酸等短链脂肪酸,有助于肠道营养物质的吸收并维持肠道健康[22]。同时,添加菌或酶的试验组的疣微菌门均高于对照组,有研究发现疣微菌门的微生物,特别是AKK菌,具有抗炎和肠道屏障功能,高丰度的疣微菌门与宿主的健康密切相关[23]。因此,添加粪肠球菌和复合酶制剂后,有利于增加0~8周龄蛋鸡盲肠有益菌的丰度,促进其肠道健康。
拟杆菌属和粪杆菌属都是肠道内常见益生菌,粪杆菌属可以促进短链脂肪酸(如丁酸)的产生,对于维持肠上皮细胞的健康和加强紧密连接增强肠道屏障功能至关重要[24]。另外,研究表明鸡盲肠中粪杆菌属的丰度与丙酸盐、丁酸盐和戊酸盐浓度相关[25-26]。本研究中,粪肠球菌和复合酶制剂协同添加后,拟杆菌属和粪杆菌属相对丰度显著高于其他组,添加粪肠球菌后阿克曼氏菌属(Akkermansia)的相对丰度也显著提高。阿克曼氏菌属下的嗜黏蛋白阿克曼氏菌(Akkermansia muciniphila)在代谢调节、免疫调节和肠道健康保护方面发挥着重要作用,被称为下一代益生菌[27]。另外,本研究中CE+EF组的标志微生物为粪杆菌属和拟杆菌属,EF组的标志微生物为阿克曼氏菌属,CE组为克里斯滕森菌属,说明饲粮中添加粪肠球菌和酶制剂可以增加0~8周龄蛋鸡肠道内有益菌的含量。
本研究中肠道菌群与表型相关性分析结果显示,蛋鸡血清GH含量与肠道内拟杆菌属和粪杆菌属相对丰度呈显著正相关,GHR含量与拟杆菌属、科林斯菌属、Intestinimonas的相对丰度呈显著正相关,GPX的含量与Firmicutes unclassified的相对丰度呈显著正相关。推测饲粮添加粪肠球菌和复合酶制剂能够通过增加0~8周龄蛋鸡肠道内有益菌的含量,改善微生物菌落结构,提高营养物质的利用率,同时影响机体内GH、GHR和GPX的分泌水平,从而促进其生长发育和肠道健康。
-
饲粮中添加粪肠球菌和复合酶制剂可以提高0~8周龄蛋鸡GH、GHR和GPX的含量,促进其生长发育和抗氧化能力,还可以改善其肠道形态发育,且酶菌协同使用效果更好,同时还能增加盲肠中有益菌如粪杆菌属、拟杆菌属、阿克曼氏菌属以及克里斯滕森菌属等的丰度,促进蛋鸡的肠道健康。





 DownLoad:
DownLoad: