-
开放科学(资源服务)标识码(OSID):

-
南方根结线虫(M. incognita)是一种专化性寄生线虫,分布广泛[1],危害严重。南方根结线虫在烟草的各个时期都可以侵染,其中在幼苗期侵染危害较大。随着连作年限的增加,土壤中线虫数量越积越多,在连作年限12年的蔬菜大棚中调查,土壤中根结线虫数量达到861.8条/100 g干土[2]。线虫在土壤中的聚集,严重危害农田作物生长。当前常用的根结线虫的防控药剂为阿维菌素(Abamectin,ABA)、噻唑膦(Fosthiazate,FST)等药剂,长期使用使线虫产生了抗性。吴青松[3]测定了南方根结线虫中抗性种群和敏感种群对噻唑膦的耐受能力,抗性种群LC50为152.18 mg/L,敏感种群LC50为55.54 mg/L,抗性种群抗性倍数是敏感种群的2.74倍。抗性种群的乙酰胆碱酯酶比活力降低,噻唑膦对线虫神经系统的抑制降低。氟吡菌酰胺对根结线虫病、烟草黑胫病具有较好的防控能力,但价格较高,生产上应用较少。武文卿等[4]发现:苜蓿田喷施5%阿维菌素乳油20 mL/667 m2会在短期内缩短蜜蜂访花时间并降低访花效率;经过1 h、2 h、4 h、24 h处理后,蜜蜂乙酰胆碱酯酶活力、羧酸酯酶活力、谷胱甘肽转移酶活力均比对照显著升高。
化学药剂作为田间主要的防治手段,常年单一使用容易使线虫形成抗性种群,为延缓抗性产生,需搭配多种化学药剂[5]或生物菌剂如淡紫拟青霉[3, 6]等进行综合防治。田间操作上,种植抗病品种[7]、作物轮作[8-9]、翻耕土地[10]等也能部分抑制根结线虫病的发生。目前也有一些植物源农药对线虫具有较好的防效,如异硫氰酸烯丙酯、黄酮类化合物杀线虫活性较高[8, 10]。周源[11]发现,万寿菊制剂、万寿菊间作、克线灵制剂防治烟草根结线虫病时,万寿菊制剂防效达到63.76%,间作达到62.00%。
香豆素类化合物作为植物源天然产物,具有抗肿瘤、抗病毒、抗氧化、降压等多种生物活性[12-13]。香豆素化合物是一类含有苯骈α-吡喃酮母核结构的芳香含氧杂环化合物,这种结构赋予其良好的生物活性和分子可塑性。香豆素类化合物来源广泛,其广泛存在于伞形科、豆科、菊科、芸香科等多种植物中。例如,伞形科的蛇床子、白芷,豆科的紫苜蓿,菊科的茵陈蒿等植物,均是香豆素类化合物的重要来源[14-16]。香豆素类化合物在松材线虫(Bursaphelenchus xylophilus)的活性研究较多,无花果中的甲醇提取物补骨脂素[17],当归中的乙酸乙酯提取物蛇床子素、二氢欧山芹醇当归酸酯、佛手柑内酯和花椒毒素[18]均对松材线虫(B. xylophilus)有较好的杀线虫活性。在狼毒根乙醇提取物的乙酸乙酯层中分离得到8种活性物质,其中狼毒素C对松材线虫72 h的LC50为2.7 μmol/L,狼毒色原酮对线虫具有更强的杀线虫活性,LC50为0.003 μmol/L [19]。
Pan等[20]对香豆素骨架中C4和C7位置进行靶向设计后,测试了衍生物对南方根结线虫(M. incognita)、腐烂茎线虫(Ditylenchus destructor)、松材线虫(B. xylophilus)和拟松材线虫(Bursaphelenchus mucronatus)的生物活性,找到4种杀线虫能力较好的衍生物,其中72 h时对南方根结线虫的LC50分别为64.0 μmol/L、37.4 μmol/L、5.1 μmol/L、42.4 μmol/L。Wang等[21]从白亮独活的根提取物中分离到3种对松材线虫具有毒杀活性的化合物分别为8-香叶草氧基补骨脂素、欧前胡素、独活内酯,72 h的LC50分别为188.3 mg/L、161.7 mg/L、114.7 mg/L。蛇床子和白芷根的乙酸乙酯提取物对松材线虫具有显著的杀线虫活性,同时抑制卵孵化,降低线虫取食和繁殖能力,也能抑制松材线虫的AChE和Ca2+-ATP酶[20]。本文从香豆素类化合物对南方根结线虫的生物活性测定出发,筛选出对根结线虫具有防控潜力的化合物,以常规药剂阿维菌素作为对照,为田间控病试验提供理论指导。
HTML
-
试验仪器:BSA124S-CW电子天平(上海齐欣科学仪器有限公司)、DHP-9162电热恒温培养箱(德国Sartorius公司)、CCT-3320纯水机(成都威思达智能科技有限责任公司)、BDS400倒置光学显微镜(重庆奥特光学仪器有限责任公司)。
试验试剂:香豆素(Coumarin)、6-甲基香豆素(6-Methylcoumarin)、7-甲基香豆素(7-Methylcoumarin)、东莨菪内酯(Scopoletin)、瑞香素(7,8-Dihydroxycoumarin)、秦皮乙素(6,7-Dihydroxycoumarin)、秦皮素(7,8-dihydroxy-6-methoxycoumarin)、6-硝基香豆素(6-Nitrocoumarin)、蛇床子素(Osthole)、滨蒿内酯(Scoparone)、7-羟基-4-甲基香豆素(4-Methylumbelliferone)。丙酮、吐温-80。
试验生物材料:带有根结的发病烟根,根据前期的研究结果,线虫种类已经鉴定为南方根结线虫,田间移栽苗为大棚育苗65~75 d的烟苗。
-
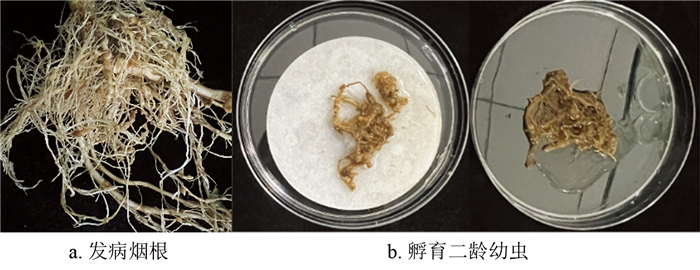
在育苗棚内选取发病严重并有大量根结的烟根(图 1),将烟根附着的土壤清洗干净后带回天然产物农药研究室。将根结剪短后放入平板培养皿中,培养皿中垫一层滤纸,加入少量RO水润湿滤纸和浸没根结,之后将平板放入25~28 ℃恒温培养箱中。孵化1~2 d后将含线虫的水溶液吸入离心管中。离心浓缩后取100 μL在96孔板进行计数,3孔计数的平均值为悬浮液中的线虫浓度,大致为50条/100 μL[22-24]。
香豆素类化合物不溶于水,借助丙酮和吐温-80使香豆素类化合物溶于水。2 000 mg/L药剂配制方法:(1)0.1%吐温-80水溶液的配制。枪头吸取10 mg吐温-80于烧杯中,加入10 mL RO水,置于超声机内超声10 min,制成0.1%吐温-80水溶液。(2)万分之一天平称取4 mg的药,置于2 mL离心管中,同时用枪头吸取4 mg吐温-80于离心管中。(3)向离心管中加入10~30 μL丙酮,快速搅拌,使香豆素类物质溶解于丙酮中,加入2 mL 0.1%吐温-80水溶液,搅拌均匀后,置于超声波内振荡10 min。
-
吸取100 μL(约50条J2)的南方根结线虫悬浮液于96孔细胞培养板中,将香豆素类化合物配制成2 000 mg/L、1 000 mg/L、500 mg/L、250 mg/L、125 mg/L、62.5 mg/L、31.25 mg/L的水溶液,各取100 μL加入盛有线虫液的96孔板内混匀。每个浓度设置8个孔位,24 h和48 h各观察4孔。每个药剂不同时间和不同批次线虫状态下进行3次试验,确保数据可再现。
试验开始时,评估0.1%吐温-80、丙酮、RO水对线虫的杀虫活性,确认无杀虫活性或活性较低。在25~28 ℃静置24 h后于倒置显微镜下统计4个孔内的线虫情况,48 h统计另外4孔。线虫死亡鉴定标准统一为“僵直不动”,为预防线虫假死,统计死亡数量时在96孔板中加入1 mol/L的NaOH 100 μL,观察线虫对刺激的反应,未对刺激做出反应则默认为死亡。计算线虫校正死亡率,计算方法如下:
-
根据初筛中125 mg/L对南方根结线虫的室内杀虫活性,选择香豆素和6-甲基香豆素作为田间试验药剂,设置100 mg/L和300 mg/L作为田间处理浓度。配制5%香豆素乳油和5%6-甲基香豆素乳油,后续按166倍稀释灌根和500倍稀释灌根。按照当地常规方式进行育苗、施肥、移栽和管理,试验共设计6个处理,每个处理设置3个小区,每小区60株烟、18个小区、1 080株烟,外围设置保护行。试验地点位于四川凉山州会理市黎溪镇横塘坝村烟草种植基地单元。该地常年种植烟草作物,前茬作物为小麦。烟草育苗时间为2024年2月21日-4月26日,烟草移栽时间为4月27日。
试验设置6个处理。C1:香豆素灌根,20 g/667 m2,100 mL;C2:香豆素灌根,60 g/667 m2,100 mL;C3:6-甲基香豆素灌根,20 g/667 m2,100 mL;C4:6-甲基香豆素灌根,60 g/667 m2,100 mL;C5:阿维菌素,800倍液灌根;CK:清水对照。实验室配制成5%香豆素和5%6-甲基香豆素乳油后,离心管分装并锡纸避光包装后带入田间。
-
按《烟草农艺性状调查测量方法》(YC/T 142—2010)标准测定农艺性状,主要包括烟株的株高、茎围、有效叶片数、最大叶长、最大叶宽,每小区测定烟株5株,根据公式计算叶面积。烟草病害发生情况按《烟草病虫害分级及调查方法》(GB/T 23222—2010)国家标准调查。结合当地的病害发生特点,主要对根结线虫病进行调查,调查每个小区的发病株数及发病级数,计算发病率,病害调查可与测定烟草农艺性状同步进行。根据根结线虫病的发生情况,在发病初期开始调查,每隔5 d调查1次,连续调查4次以上。
在公式中,Xi和Xi-1分别表示ti和ti-1的病情指数,ti-ti-1代表后一次调查时间与前一次调查时间的间隔。
-
依据生物活性测定试验的校正死亡率及其药剂浓度对数,使用Excel 2016软件得出毒力回归方程y=a+bx,用SPSS 27.0计算供试药剂的LC50及其相应置信区间。田间试验中用Duncan新复极差法对不同处理进行多重比较。用Origin 21进行试验数据绘图。不同小写字母表示差异显著(p<0.05)。
1.1. 试验材料
1.2. 试验设计
1.2.1. 二龄幼虫悬浮液准备
1.2.2. 室内试验处理
1.3. 田间试验验证
1.4. 农艺性状和病害调查
1.5. 数据分析
-
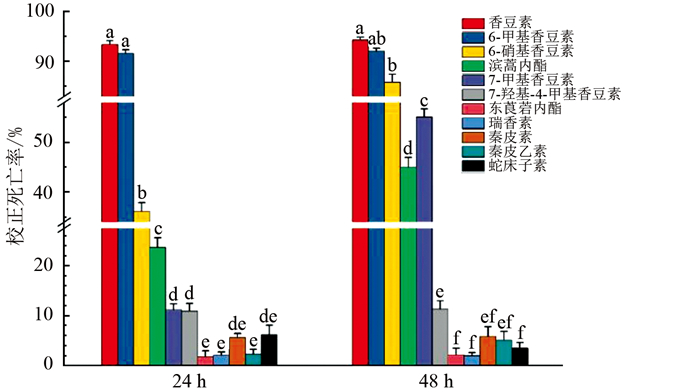
在现有的11种香豆素化合物在125 mg/L条件下对南方根结线虫的杀虫活性测试如图 2所示,阴性对照和RO水(空白对照)死亡率均低于5%。多数香豆素类化合物,如7-羟基-4-甲基香豆素、秦皮乙素、秦皮素、东莨菪内酯在1 000 mg/L时24 h和48 h对线虫致死率较低,不符合预期。500 mg/L瑞香素在24 h时对线虫校正死亡率仅为43.03%,500 mg/L蛇床子素在24 h时对线虫致死率为0。测试发现,香豆素、6-甲基香豆素、6-硝基香豆素在125 mg/L对线虫具有较好的杀虫活性,香豆素和6-甲基香豆素在125 mg/L、24 h时对南方根结线虫具有90%以上的校正死亡率,与其他化合物存在显著性差异。6-硝基香豆素和7-甲基香豆素24 h时杀虫活性较低,48 h时校正死亡率分别为85.77%和55.06%。滨蒿内酯在125 mg/L时对线虫有一定致死率,但效果较差,48 h校正死亡率仅为45.00%,其他药剂在125 mg/L时对南方根结线虫的杀虫活性较低,24 h和48 h内均未表现出较好的杀线虫活性。据此,选择125 mg/L时生物测定活性较好的香豆素和6-甲基香豆素进行复筛。
-
根据前期的香豆素药剂杀线虫活性评价发现,在125 mg/L时,香豆素和6-甲基香豆素对根结线虫均具有较好的杀虫活性。在3.75 mg/L、7.5 mg/L、15 mg/L、30 mg/L、60 mg/L、120 mg/L等6个浓度梯度下筛选对线虫的致死率,并与常规药剂阿维菌素对比,其LC50、毒力回归方程等如表 1所示。
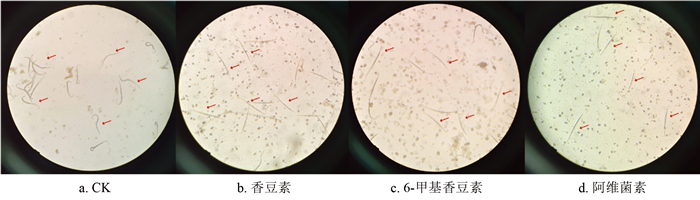
120 mg/L香豆素和6-甲基香豆素处理24 h后线虫的生存状况(如图 3所示),在RO水中,线虫活性较高,经香豆素、6-甲基香豆素、阿维菌素处理后,加入1 mol/L的NaOH刺激,线虫僵直,说明经香豆素、6-甲基香豆素、阿维菌素处理后的线虫已经死亡。
-
对植株旺长期的农艺性状调查发现(表 2),处理C1株高显著提升为60.61 cm,CK株高52.74 cm,处理C1与CK存在显著性差异,提升了14.92%;处理C1、C2、C3植株的最大叶长显著提升,与CK存在显著性差异,分别为76.00 cm、76.00 cm、76.07 cm,提升了6.35%、6.35%、6.45%;其余的农艺性状与CK差异不显著。
-
对植株打顶7 d的农艺性状调查发现(表 3):处理C1提高了植株多项农艺性状,株高、茎围、有效叶片数分别为90.38 cm、12.44 cm、21.33片,CK为79.70 cm、11.73 cm、18.00片,与对照均存在显著性差异,分别提升了13.40%、6.05%、18.33%,其他处理差异不显著;处理C2和处理C3最大叶长显著增加,分别为80.65 cm、79.97 cm,CK仅为74.30 cm,提升了8.55%、7.63%;处理C2还显著增加了植株的最大叶面积为1 556.82 cm2,CK为1 389.33 cm2,提升了12.06%。
-
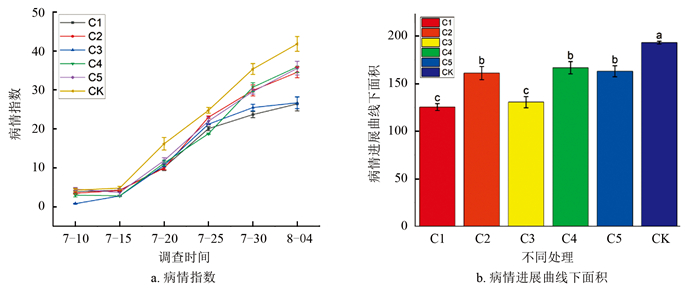
2024年7月初进入根结线虫发病期。7月10日调查(图 4a),处理C3和C4可以有效抑制病情指数,处理C3和C4病情指数最低为0.78和2.96,CK为4.29,防效为81.82%和31.00%。7月20日调查,处理C3和C4病情指数最低为9.95和10.09,CK病情指数为16.17,二者与CK存在显著性差异,防效分别为38.47%和37.60%。8月4日调查末期,发现处理C1和C3可以有效抑制病情进展,在所有对照和处理中病情指数最低,为23.69和25.48,对照为41.83,相关防效为43.37%和39.09%。
基于7月30日到8月4日的病情进展曲线下面积表明(图 4b),处理C1、C3分别为125.27和130.48,CK为193.00,与CK间存在显著性差异,处理C1和C3延缓了植株的病情进展。
2.1. 11种香豆素类化合物的室内杀虫活性比较
2.2. 香豆素和6-甲基香豆素对二龄幼虫生物活性测定拟合方程
2.3. 不同处理对烟草农艺性状的影响
2.3.1. 不同处理对植株旺长期农艺性状的影响
2.3.2. 不同处理对植株打顶期农艺性状的影响
2.4. 不同处理对烟草根结线虫病的防治效果
-
本试验评价了11种常见香豆素类化合物对南方根结线虫的致死率,发现香豆素和6-甲基香豆素在125 mg/L时可以有效杀死线虫,校正死亡率达到90%以上。在大田试验中,处理C1可以有效提升植株旺长期的株高、最大叶长,在打顶期仍然能维持株高、茎围、有效叶片数优势。在调查末期,处理C1和处理C3在所有对照和处理中病情指数最低,为23.69和25.48。
南方根结线虫作为一种根系寄生物种,只有依赖植株才能完成整个的生命历程。烟草作为一种经济作物,根系容易遭受线虫侵害,当前市场上的阿维菌素、噻唑膦等药剂,常年使用容易产生抗药性[25-26],同时聚集在土壤中不易分解,加剧土壤酸化[27]。6-甲基香豆素对苹果树腐烂病菌(Valsa mali)和疏忽拟盘多毛孢菌(Pestalotiopsis neglecta)的抑菌活性分别为94.6%和76.9%[28]。香豆素类化合物作为植物分泌的苯丙烷类衍生物,不仅具有直接抑制作物病原体的活性,还被证实可调控微生物组结构而间接增强植物系统性抗性。在香豆素诱导的化感胁迫下,有益菌群在多花黑麦草(Lolium multiflorum)根际定植[29]。香豆素可通过调控鞘脂单胞菌属(Sphingomonas)和链霉菌属(Streptomyces)的丰度,激活其合成几丁质酶或抗生素的基因簇[30-31],香豆素可通过干扰微生物群体感应系统,抑制革兰氏阴性细菌生物膜形成[32-33]。Rastija等[34]研究了9种香豆素类衍生物对尖孢镰刀菌(Fusarium oxysporum)、禾谷镰孢菌(Fusarium culmorum)、菜豆壳球孢菌(Macrophomina phaseolina)和核盘菌(Sclerotinia sclerotiorum)的抗菌活性,发现这些衍生物可通过抑制内切-1,4-β-葡聚糖酶Ⅰ和果胶酶等细胞壁降解酶的活性而干扰真菌的侵染过程。部分香豆素类化合物能诱导植株抗性基因的表达[35-37],干扰细菌的群体感应[38]。总之,利用香豆素的杀虫能力和对土壤病原菌的抑制能力,可促进烟株健康生长。




 DownLoad:
DownLoad: