-
开放科学(资源服务)标识码(OSID):

-
粒型是影响水稻产量和品质的重要因素之一,研究水稻粒型对提高稻谷产量、改善加工品质、满足不同消费者的需求等有重要作用。粒型是数量性状,其调控机制复杂,受遗传背景影响较大。尽管目前已鉴定和克隆到多个粒形基因,但其调控网络仍不完善,在育种工作中的应用较少。挖掘更多的粒型数量性状基因座(Quantitative Trait Locus,QTL)可为完善水稻粒型调控机制奠定基础,同时为培育优质高产的水稻新品种提供基因资源。
目前在水稻中已发现有400多个与粒型相关的QTL,主要在G蛋白信号传导、泛素蛋白酶体降解、丝裂原活化蛋白激酶(Mitogen-Activated Protein Kinase,MAPK)信号通路、转录因子调控、植物激素调控等途径中发挥作用[1]。
蛋白质泛素化过程是由泛素激活酶E1、泛素结合酶E2和泛素连接酶E3等级联催化完成的[2]。目前已克隆的通过泛素蛋白酶体途径调控粒型的基因有GW2 [2]、TUD1 [3]、WTG1/OsOTUB1 [4]、LG1/OsUBP15 [5]、HDR3 [6]等。GW2是首个被克隆的水稻QTL,编码一种泛素连接酶E3[2]。研究发现GW2可以将WG1泛素化并促进其降解,而WG1可以抑制粒宽和粒质量的负调控转录因子OsbZIP47的转录激活活性[7]。
G蛋白由Gα、Gβ和Gγ 3个亚基构成,通过调控植物信号转导过程影响植株的生长和发育[8]。在水稻中共鉴定到1个Gα基因(RGA1),1个Gβ基因(RGB1)和5个Gγ基因(RGG1、RGG2、GS3、DEP1、GGC2),均参与水稻粒型调控[8]。有研究表明5个Gγ亚基都可以与AP2家族转录因子GR5相互作用,通过调控下游靶基因的表达控制颖壳细胞数目和大小决定水稻籽粒大小[9]。
MAPK信号通路是由MAPK、MAPKK和MAPKKK 3种不同类型的激酶组成的三级激酶级联反应,目前已克隆的涉及MAPK信号途径调控水稻粒型的基因有OsMKK4/SMG1、OsMAPK6、OsMKKK10 [10]、OsMKP1/GSN1 [11]、OsMKKK70 [12]等。OsMKKK10-OsMKK4-OsMAPK6[10]和OsMKKK70-OsMKK4-OsMAPK6[12]级联反应在水稻籽粒大小调控中发挥重要作用。
转录因子在水稻粒型调控中有着关键作用。目前已发现的调节水稻粒型的转录因子有GS9 [13]、OsSNB [14]、GL6 [15]、OsSPL2 [16]、DLT2 [17]等。DLT2是植物特异性转录因子GRAS成员,在BR信号传导系统中起正调控作用,可以调控水稻株型和籽粒大小[17]。
植物激素在水稻粒型形成和调控中同样起着至关重要的作用,目前已挖掘出许多与调控水稻粒型的植物激素相关的基因。与生长素相关的水稻粒型调控基因有OsIAA3、OsARF25、Gnp4/LAX2 [18]、OsARF6、OsAux3 [19]、OsTI bR1、OsIAA10、OsARF4 [20]、TGW3 [2-21]等。TGW3是籽粒大小的负向调节因子,可以磷酸化修饰一个典型的Aux/IAA蛋白OsIAA10,OsIAA10的磷酸化促进其与OsTIR1的相互作用,但会阻碍其与OsARF4的相互作用,TGW3通过改变生长素遗传调控模板OsTIR1-OsIAA10-OsARF4的信号,进而影响水稻籽粒大小[20-21]。调控水稻粒型的生长素模块还有OsIAA3-OsARF25-SMOS1[18]、OsIAA3-OsARF25-OsERF142[18]和miR167a-OsARF6-OsAUX3[19]等。通过油菜素内酯合成途径调控水稻粒型的相关基因有D11 [22]、BRD1 [23]、BRD2 [24]等。油菜素内酯信号传导途径中调控水稻粒型的基因有OsBZR1 [25]、GW5/GSE5 [26]、qGL3、OsGSK3 [27]、OFP3 [28]等。GW5在油菜素内酯信号传导过程中起正调节作用,可与GSK2互作并抑制GSK2的自磷酸化,以此抑制GSK2对DLT和OsBZR1的磷酸化作用,导致未被磷酸化的DLT和OsBZR1蛋白在细胞核中积累,从而调控油菜素内酯响应基因的表达水平,最终影响水稻的生长[26]。
水稻育种目标是获得理想的品种,利用分子设计育种将优良粒型基因进行聚合,可以定向改良粒型,满足消费者偏好。水稻粒型属于数量性状,受多基因共同调控,遗传机制复杂。染色体片段代换系(Chromosome Segment Substitution Line,CSSL)是仅含少量来自供体基因组代换片段的纯合近等基因系材料,除代换片段外,其他遗传背景与受体亲本完全一致,所以CSSL可以将复杂的数量性状分解为单个遗传因子,提高QTL定位的准确性,为设计育种作好充足的准备[29]。单片段代换系(Single Segment Substitution Lines,SSSL)仅含有来自供体基因组的1个代换片段,从而使复杂的数量性状被分解到单个代换片段上,成为“质量基因”,因此SSSL可以成为QTL精细定位及分子设计育种的绝佳材料[29]。
本研究鉴定到一个水稻6片段染色体代换系Z796,该代换系以西南地区优良籼型恢复系西恢18作为受体、粳型旱稻沪旱3号为供体构建而成,再以西恢18/Z796构建的次级F2代分离群体为材料对粒型等农艺性状进行QTL定位,根据初定位结果进一步进行次级代换系的选育及粒型等QTL的验证和遗传效应分析。相关研究结果为水稻粒型基因的精细定位及新一代的分子设计育种奠定了良好理论基础。
HTML
-
CSSL-Z796是用西南地区优良籼型恢复系西恢18作为受体亲本、粳型旱稻沪旱3号作为供体亲本进行杂交后再经过多代的回交及自交,同时结合全基因组多态性SSR分子标记辅助选择(Molecular Marker-assisted Selection,MAS)育成的短宽粒代换系。
以西恢18和CSSL-Z796杂交构建的由100个单株组成的次级F2代群体作为QTL定位群体。
根据QTL定位结果,从F2代群体中选择携带目标QTL的单株作为次级片段代换系材料。
-
在覆盖水稻全基因组的429个SSR标记的基础上,共筛选到西恢18与沪旱3号之间的多态性标记263个。之后利用筛选到的多态性标记从BC2F1代进行MAS并结合表型观察,最终在BC3F6代选育出短宽粒表型的含有6个代换片段的CSSL-Z796。染色体的鉴定方法参照文献[30]的描述,当某个多态性标记的基因型鉴定结果与西恢18(A)一致时,认为该段DNA未被供体亲本替代,仍是西恢18基因组; 当该标记的基因型鉴定结果与沪旱3号(B)一致时,则认为该段DNA被供体亲本沪旱3号的基因组所代替。B标记的连续区间表示代换片段,估计代换片段长度的计算方法参照文献[31]。
-
水稻植株成熟后,平地面收割Z796和西恢18各10株、F2代群体共100株,依据《农作物田间试验记载项目及标准》进行表型考察及数据统计。考察各个植株的高度、有效穗数、穗长、一次枝梗数、二次枝梗数、每穗总粒数、每穗实粒数、结实率、粒长、粒宽、长宽比、千粒质量、单株产量共13个性状,具体测定方法参照文献[32],最后使用Microsoft Excel 2010对各性状参数进行统计分析。
-
在灌浆期取幼嫩的植株籽粒,利用扫描电镜在-20 ℃条件下对西恢18和Z796的籽粒颖壳外表皮细胞进行观察和数据统计分析[33]。
-
用改良的CTAB法[34]提取西恢18、Z796和100个F2代群体单株的DNA,参照文献[32]描述的方法进行PCR扩增,再用10%的非变性聚丙烯酰胺凝胶电泳对100个F2代群体单株进行基因型鉴定。对西恢18基因型、Z796基因型、杂合基因型、缺失基因型分别赋值-1、1、0和·。将100个F2代群体单株的基因型值及对应的表型值结合,使用限制性最大似然(REML)法[32]在SAS(V9.3 SAS Institute Inc,Cary,NC,USA)统计软件上进行计算,以此定位QTL,以p<0.05为阈值来判断是否存在QTL。
-
根据F2代群体QTL初定位的结果,结合100个F2代群体单株的基因型和表型,从中选择14个单株种成14个株系,植株成熟后再收割供、受体亲本及每个株系20株,利用MAS进一步选育含有目标QTL的单/双片段代换系。根据F3代群体的基因型和表型,又从14个株系中选择了24个单株种成株系,在F4代群体再次进行单/双片段代换系的选育。
-
F3和F4代群体共鉴定到6个SSSL(S1-S6),收获西恢18、Z796各10株,所有S1-S6植株参照1.2.2描述的方法测量粒长、粒宽等农艺性状。与受体亲本的染色体相比,每个SSSL只有1个代换片段存在差异,从遗传方面解释,其表现出的和受体亲本有差异的表型均与这个差异代换片段有关。对于每个SSSLi(i=1、2、…、6),先提出无效假设H0,即SSSLi(i=1、2、…、6)的代换片段不存在调控某种性状的QTL。针对这个性状,利用IBM SPSS Statistics 25.0的ONE-WAY ANOVA和LSD对每个SSSLi和西恢18进行多重比较统计分析,如果p<0.05,则否定无效假设H0,认为SSSLi的代换片段存在控制某种性状的QTL。QTL的加性效应通过各个SSSLi和西恢18该种性状表型值差异的一半计算[35-36],所有统计计算均在Microsoft Excel 2010中完成。
-
植株成熟后,同样收割所有双片段植株,参照1.2.2的方法测量粒长等性状。应用T检验来检测各个性状表型值西恢18+DSSLab和SSSLa+SSSLb之间的显著性。其中:DSSLab表示含有a、b两个代换片段的DSSL的某个性状的表型值; SSSLa、SSSLb分别表示含有a、b代换片段的SSSL的某个性状的表型值; 西恢18代表西恢18某个性状的表型值。以此判定QTL间是否存在上位性效应,如果p<0.05,则认为QTL间存在上位性效应。DSSL QTL间的上位性效应以[(西恢18+DSSLab)-(SSSLa+SSSLb)]/2估计[35, 37],所有统计分析均在Microsoft Excel 2010中进行。
1.1. 试验材料
1.2. 试验方法
1.2.1. Z796代换片段鉴定
1.2.2. 表型分析和农艺性状调查
1.2.3. 细胞学分析
1.2.4. QTL定位
1.2.5. 单/双片段代换系的选育
1.2.6. 基于单片段代换系(SSSL)QTL的加性效应分析
1.2.7. 基于双片段代换系(DSSL)QTL的上位性效应分析
-
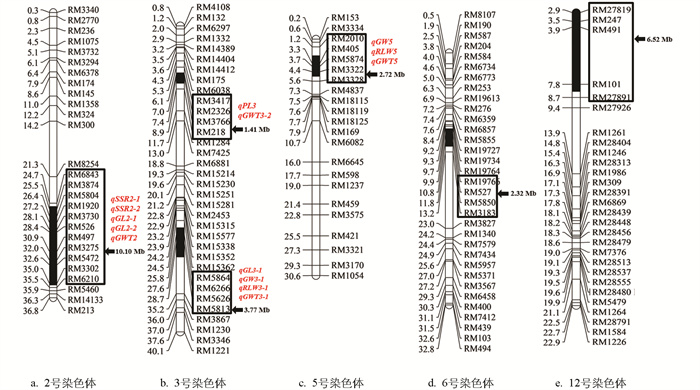
对10株Z796进行代换片段的鉴定及遗传背景检测,10株Z796的鉴定结果一致,鉴定到Z796共携带来自沪旱3号的6个染色体代换片段,分别位于水稻第2、3、5、6、12号染色体上,除3号染色体上有2个代换片段外,其余染色体上均只含1个代换片段。代换片段总长度共有26.84 Mb,最小代换片段长度有1.41 Mb,最大代换片段长度有10.10 Mb,代换片段平均长度为4.47 Mb(图 1)。
-
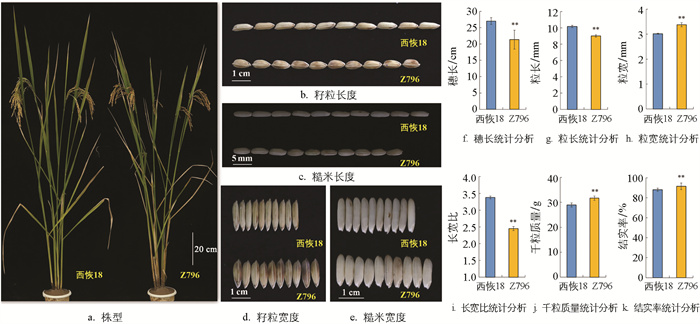
与西恢18相比,Z796的籽粒宽度(图 2d、e、h)、结实率(图 2k)和千粒质量(图 2j)均极显著增加,粒宽增加了1.14 mm、结实率增加了3.48%、千粒质量增加了2.74 g; 而粒长(图 2b、c、g)、长宽比(图 2i)和穗长(图 2f)则极显著降低,粒长减少了1.16 mm、长宽比减少了0.93、穗长减少了5.71 mm。株高、一次与二次枝梗数、每穗实粒和总粒数、有效穗数及单株产量的差异没有统计学意义。
-
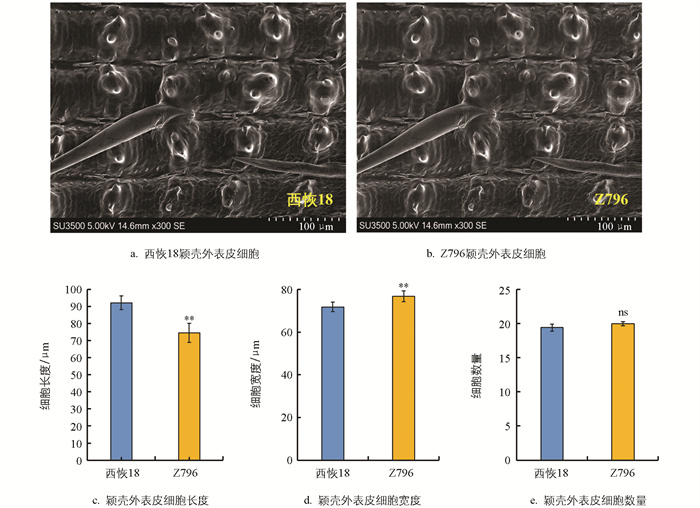
为了探究Z796籽粒长度减少、宽度增加的原因,利用扫描电镜观察了受体亲本西恢18与Z796灌浆期的颖壳外表皮细胞。结果显示,与西恢18相比,Z796颖壳外表皮的细胞长度极显著减少,平均减少了17.64 μm(图 3c); 细胞宽度极显著增加,平均增加了5.04 μm(图 3d); 相同面积内,颖壳外表皮细胞数量无显著变化(图 3e)。结果暗示Z796籽粒长度减少、宽度增加可能是由颖壳外表皮细胞横向扩张引起的。
-
对结合西恢18和Z796构建的F2代群体中单株基因型和表型数据进行QTL定位,共定位到14个QTL,涵盖粒长、粒宽、长宽比、穗长、结实率、千粒质量共6个性状。其中有13个QTL为贡献率大于10%的主效QTL,只有qPL3的贡献率小于10%,为8.35%,qGW5的贡献率最大,为79.68%(表 1)。
共鉴定到3个粒长QTL:qGL2-1、qGL2-2、qGL3-1。其中:qGL3-1的加性效应为负,导致粒长减少了0.41 mm,贡献率为50.67%; qGL2-1、qGL2-2的加性效应为正,分别增加粒长0.15 mm、0.22 mm,贡献率分别为13.31%、29.43%。共鉴定到2个粒宽QTL:qGW3-1、qGW5,加性效应均为正,分别增加粒宽0.08 mm、0.20 mm,贡献率分别为12.68%、79.68%。共鉴定到2个长宽比QTL:qRLW3-1、qRLW5,加性效应均为负,分别使长宽比减少0.12、0.11,贡献率分别为38.93%、37.09%。共鉴定到2个结实率QTL:qSSR2-1、qSSR2-2。qSSR2-1的加性效应为负,导致结实率降低7.19%,贡献率为13.00%; qSSR2-2的加性效应为正,增加结实率13.95%,贡献率24.11%。共鉴定到4个千粒质量QTL:qGWT2、qGWT3-1、qGWT3-2、qGWT5。其中:qGWT3-1的加性效应为负,导致千粒质量减少1.55 g,贡献率为39.40%; 其余3个加性效应均为正,分别增加千粒质量2.12 g、1.03 g、1.36 g,贡献率分别为55.60%、28.72%、38.29%。鉴定到1个穗长微效QTL:qPL3,加性效应为负,导致穗长减少0.73 cm(表 1)。
-
在QTL初定位的基础上,通过MAS法在F3和F4代群体中分别选育SSSL(S1-S4)、(S1-S6)和DSSL(D1-D4)、(D1-D6)。SSSL的S1-S2、S3、S4-S5和S6分别含有染色体3、染色体5、染色体6和染色体12上的代换片段。DSSL的D1含有染色体3和染色体5上的代换片段; D2含有染色体3和染色体6上的代换片段; D3含有染色体3和染色体5上的代换片段; D4含有染色体3和染色体6上的代换片段; D5含有染色体5和染色体6上的代换片段; D6含有染色体6和染色体12上的代换片段(表 2,图 4)。
-
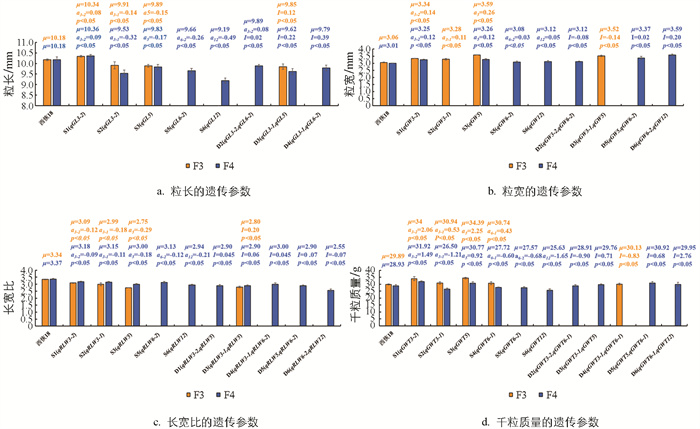
F3代群体鉴定到的SSSL共检测到16个QTL,F4代群体共检测到23个QTL。两年同时检测到的QTL有12个。F3和F4代群体的SSSL(S1-S6)进一步验证了F2代群体9个QTL(qGL3-1、qGW3-1、qGW5、qPL3、qRLW3-1、qRLW5、qGWT3-1、qGWT3-2、qGWT5)的遗传稳定性,其中F2、F3、F4代群体均检测到的QTL有7个(qGL3-1、qGW5、qRLW3-1、qRLW5、qGWT3-1、qGWT3-2、qGWT5),这7个QTL中,除qGWT3-1外,其余QTL在3代中均出现同方向的表型值(表 1、表 2),验证了主效QTL的遗传稳定性,而qGWT3-1的遗传可能受环境影响较大。此外,有13个QTL(qGL3-2、qGL5、qGL6-2、qGL12、qGW3-2、qGW6-2、qGW12、qRLW3-2、qRLW6-2、qRLW12、qGWT6-1、qGWT6-2、qGWT12)未在F2代群体中被定位到,而通过对应的次级SSSL:S1、S3、S4、S5、S6定位到(表 2),说明SSSL具有更高的QTL检测效率。因未能选到所有对应的SSSL,部分由次级F2代群体定位的QTL无法被验证。
-
在F3和F4代群体DSSL均鉴定到携带qGL3-1和qGL5的DSSL:D3。qGL3-1(a=-0.14)和qGL5 (a=-0.15)在F3代群体互作产生了0.12的上位性效应,据DSSL的遗传模型,D3粒长遗传效应为-0.17,小于qGL3-1和qGL5的加性效应,因此D3的粒长(9.85 mm)比含有qGL3-1的S2(9.91 mm)和含有qGL5的S3(9.89 mm)小,表明qGL3-1与qGL5聚合会产生更短的籽粒。但F4代的qGL3-1 (a=-0.32)和qGL5 (a=-0.17)D3互作产生了0.22的上位效应,使D3粒长遗传效应为-0.27,介于qGL3-1和qGL5的加性效应之间,因此该年D3的粒长(9.62 mm)大于S2(9.53 mm)小于S3(9.83 mm)(图 4a)。
未有两代均检测到携带粒宽QTL的DSSL。F3代检测到1个携带粒宽QTL的DSSL:D3。F4代检测到3个携带粒宽QTL的DSSL:D2、D5、D6,均携带qGW6-2 (a=0.03),此外还分别携带了qGW3-2 (a=0.12)、qGW5 (a=0.12)、qGW12 (a=0.05)。qGW6-2与另外3个粒宽QTL互作分别产生了-0.08、0.02、0.20的上位性效应,使得D2、D5、D6的粒宽遗传效应分别为0.07、0.17、0.28。D2的粒宽遗传效应介于qGW6-2和qGW3-2的加性效应之间,因此D2的粒宽(3.12 mm)大于携带qGW6-2的S5(3.08 mm),小于携带qGW3-2的S1(3.25 mm)。D5、D6的遗传效应均大于携带对应QTL的加性效应,因此D5、D6的粒宽均大于携带对应QTL的SSSL,说明同一QTL与不同QTL聚合会产生不同的遗传效应,如聚合qGW6-2和qGW3-2未能产生更宽的籽粒,聚合qGW6-2和qGW5、qGW6-2和qGW12能产生更宽的籽粒(图 4b)。
在F3和F4代均鉴定到D3携带qRLW3-1和qRLW5。qRLW3-1 (a=-0.18)和qRLW5 (a=-0.29)在F3代互作产生了0.02的上位性效应,使得D3粒长遗传效应为-0.45 mm,小于qRLW3-1和qRLW5的加性效应,因此D3的长宽比(2.80)大于携带RLWL3-1的S2(2.99)和携带qRLW5的S3(2.75)。F4代qRLW3-1 (a=-0.11)和qRLW5 (a=-0.18)互作产生了0.06的上位性效应,该年D3粒长遗传效应为-0.23 mm,仍小于qRLW3-1和qRLW5的加性效应,同样D3的长宽比(2.90)仍小于S2(3.15)和S3(3.00)(图 4c)。
未有两年均检测到携带千粒质量QTL的DSSL。F3代检测到1个携带千粒质量QTL的DSSL:D4。F4代共鉴定到4个携带千粒质量QTL的DSSL:D2、D3、D5、D6,其中D2、D5、D6均携带qGWT6-1(a=-0.60),此外还分别携带了qGWT3-2 (a=1.49)、qGWT5 (a=0.92)、qGWT12 (a=-1.65),分别互作产生了-0.90、0.68、2.76的上位性效应,D2、D5、D6的千粒质量遗传效应分别为-0.01、1.00、0.51,其中D2千粒质量遗传效应介于qGWT3-2 (a=1.49)和qGWT6-1的加性效应之间,因此D2的千粒质量(28.91 g)大于携带qGWT6-1的S4(27.72 g),小于携带qGWT3-2的S1(31.92 g)。D5、D6的千粒质量遗传效应大于携带对应QTL的加性效应,因此D5、D6的粒宽均大于携带对应QTL的SSSL(图 4d)。
2.1. 水稻Z796的代换片段鉴定
2.2. Z796的农艺性状分析
2.3. Z796的颖壳细胞学分析
2.4. Z796代换片段携带的重要农艺性状QTL定位
2.5. 次级片段代换系的选育及粒型等QTL的验证和遗传分析
2.5.1. 次级单/双片段代换系选育
2.5.2. 基于F3和F4代群体SSSL(S1-S6)的粒型相关性状的QTL验证及加性效应分析
2.5.3. 基于F3和F4代群体DSSL目标QTL间上位性效应的比较分析
-
水稻产量构成因素包括有效穗数、每穗粒数、结实率和千粒质量。本研究鉴定到一个短宽粒表型的6片段CSSL-Z796。由图 2可知,与西恢18相比,Z796的籽粒长度减少,宽度增加,但千粒质量仍增加了2.74 g,表明粒宽增加对千粒质量的影响大于粒长减少对千粒质量产生的影响,尤其是qGW5,贡献率高达79.68%,可应用于水稻的育种中。
在QTL初定位的基础上,通过MAS法在F3代和F4代中分别选育SSSL(S1-S4)、(S1-S6)。SSSL仅含有1个来自供体亲本的代换片段,可以将复杂的数量性状分解为单个质量基因,进一步纯化遗传背景,提高QTL的检测效率[29]。本研究在F3代和F4代的SSSL中分别检测到16个和23个QTL,比F2代鉴定到的QTL更多,验证了染色体SSSL QTL检测效率更高。且SSSL可以鉴定出单个数量基因的遗传效应,是水稻粒型遗传研究和分子设计育种的良好材料。
-
在初定位中共鉴定到14个QTL,包括2个粒宽、3个粒长、2个长宽比、2个结实率、4个千粒质量、1个穗长QTL。qGW5、qRLW5、qGWT5位于同一染色体区间,已报道的GW5 [26]、GS5 [38]、OsDER1 [39]均位于该区间。已报道的GW5通过参与油菜素内酯信号传导负调控水稻粒宽[26]。GS5是水稻籽粒大小的正向调节因子,其通过抑制OsBAK1和OsMSBP1的相互作用增加籽粒大小[38]。OsDER1的抑制或过表达都会导致水稻种子变小[39]。粒长qGL3-1和千粒质量qGWT3-2位于同一区间,已报道的qGL3 [27, 40]、GF14f [41]均位于该区间。qGL3编码一个蛋白磷酸酶(OsPPKL1),其负调控粒长和千粒质量[27, 40]; GF14f负调控籽粒发育和灌浆[41]。粒长qGL2-2与已报道的GL2 [42]; PGL2 [43]; AFG1 [44]位于同一区间。GL2是OsGRF4的等位基因,通过编码一种生长调节因子正调控水稻穗长和粒型[42]; PGL2编码一个非典型bHLH蛋白,正向调节籽粒长度和重量[43]; AFG1可能通过调控颖壳细胞的扩张和增殖负调控水稻籽粒大小[44]。qGWT3-1所在区间内已报道的千粒质量基因有LPA1 [45]。qGWT2区间内已报道的千粒质量基因有OsVPE3 [46]。结实率qSSR2-1与已报道的基因OsGL1-4在同一区间[47]。结实率qSSR2-2、粒长qGL2-2与粒长基因AFG1 [44]位于同一区间。穗长qPL3与已报道的穗长基因D88 [48]、OsAPC6 [49]、pls2 [50]、OspPLAIIIα [51]位于同一区间。其他QTL- qGL2-1、qGW3-1、qRLW3-1区间内未见已报道的基因。
-
植株的表型是基因与环境共同作用的结果,一个植株的某个表型值可表示为:表型值=基因型值+环境偏差+基因与环境的互作效应。基因型值受基因的加性效应、显性效应和上位性效应共同影响[52]。在解析单个QTL遗传效应的基础上进一步分析不同QTL间的上位性效应可以解析复杂性状的遗传效应,为目标QTL的聚合育种打好基础。
本研究在F3代和F4代中同时检测到1个DSSL D3,分析了不同年份粒长(qGL3-1、qGL5),长宽比(qRLW3-1、qRLW5)QTL间的上位性效应。F3代和F4代聚合qGL3-1和qGL5的D3粒长上位性效应方向均一致,聚合qRLW3-1和qRLW5的D3两年的上位性效应方向均一致,验证了其遗传的稳定性。虽然通常不同年份相同QTL聚合产生的上位性效应方向一致,但最终籽粒表型效果却存在差异。如F3代分析结果表明qGL3-1 (a=-0.14)和qGL5 (a=-0.15)聚合的D3产生了0.12 mm的上位性效应,但D3的遗传效应却小于qGL3-1和qGL5的加性效应,该年聚合qGL3-1和qGL5产生更短的籽粒。而F4代中qGL3-1 (a=-0.32)和qGL5 (a=-0.17)聚合产生了0.22 mm的上位效应,D3的粒长遗传效应介于qGL3-1和qGL5的加性效应之间,最终D3粒长位于含qGL3-1的S2和含qGL5的S3之间。
两个调控产量性状的正向(负向)QTL聚合,往往产生的上位性效应是负向(正向)的,至于是否会导致该性状变得更大(更小),取决于两个QTL的加性效应及二者的上位效应代数和的绝对值与单个QTL最大加性效应绝对值的差。如果是正值,则会导致该性状变得更大(更小); 如果是负值,则不会导致该性状变得更大(更小)。且不同年份间、遗传与环境效应间的互作会对表型值产生影响,所以最终的聚合效果存在差异。在了解单个基因的加性效应和之间的互作效应关系的基础上,结合环境影响因素,可对已知基因型的植株表型进行预测,提前预测设计育种能产生的效果。
-
为了挖掘更多的粒型QTL并构建西南地区覆盖水稻全基因组的籼型亲粳单片段代换系文库,本研究鉴定了以西南地区优良籼型恢复系西恢18为受体亲本、粳型旱稻沪旱3号为供体亲本构建的6片段CSSL-Z796,利用西恢18和Z796构建的次级F2代分离群体作为QTL定位材料,共鉴定到14个调控水稻粒型等农艺性状的QTL,贡献率为8.36%~79.68%。于F3和F4代群体中分别选育了单片段代换系(S1-S4)、(S1-S6),双片段代换系(D1-D4)、(D1-D6)。利用F3及F4代中选育出的单片段代换系进行QTL复定位,共有7个QTL(qGL3-1、qGW5、qRLW3-1、qRLW5、qGWT3-1、qGWT3-2、qGWT5)在F2、F3、F4代中被重复检测到,且除qGWT3-1外,其余QTL均出现同方向的表型值。相同QTL在不同年份聚合也会产生同方向的上位性效应,如qGL3-1和qGL5、qRLW3-1和qRLW5在不同年份聚合产生的上位性效应方向一致,说明主效QTL的遗传较稳定。同一QTL与不同QTL聚合会产生不同的遗传效应,如聚合qGW6-2和qGW3-2未能产生更宽的籽粒,聚合qGW6-2和qGW5、qGW6-2和qGW12能产生更宽的籽粒。挖掘鉴定不同QTL并探索QTL间的遗传效应,为未来设计育种提供理论基础,具有重要意义。





 DownLoad:
DownLoad: